Efficacy Comparison: Phentermine vs. Ozempic (Semaglutide) Body Weight Loss
Phentermine is an inexpensive, fast-acting appetite suppressant approved since 1959, but it is FDA-labeled for short-term use only (a few weeks) and carries cardiovascular contraindications that rule it out for many patients. Ozempic (semaglutide) costs far more but produces roughly double the weight loss over the long term per clinical trial data, and is appropriate for sustained treatment. Your ideal choice depends on your cardiac health, budget, how much weight you need to lose, and whether you have type 2 diabetes.
What Is Phentermine?
Phentermine is a Schedule IV controlled substance that has been FDA-approved for weight management since 1959, making it one of the oldest weight-loss medications still in clinical use. It remains among the most widely prescribed weight-loss drugs in the United States due to its low cost and rapid onset.
How Phentermine Works
Phentermine is a sympathomimetic amine, structurally similar to amphetamine, that works primarily by triggering the release of norepinephrine in the hypothalamus. This produces appetite suppression and a mild increase in metabolic rate. Per the phentermine HCl prescribing information on DailyMed, the anorectic effect operates through central nervous system stimulation, which is both the drug’s main benefit and the source of most of its risks.
Unlike GLP-1 agonists, phentermine does not slow gastric emptying, does not improve blood sugar control, and has no cardiovascular benefit. It is a pure appetite suppressant with stimulant properties.
Dosage and Duration
The standard adult dose is 37.5 mg once daily (as tablets or capsules). Per the Suprenza FDA prescribing information, phentermine is indicated only as short-term monotherapy (a few weeks) for the management of exogenous obesity. The label explicitly notes that tolerance to the anorectic effect typically develops within a few weeks, at which point the drug should be discontinued. The commonly cited “up to 12 weeks” guidance comes from clinical practice and older labeling conventions; the current FDA label says “a few weeks.”
Phentermine/topiramate extended-release (brand name Qsymia) is a separate combination product that delivers a lower phentermine dose alongside an anticonvulsant, it is not the same as standalone phentermine and carries its own distinct efficacy, safety, and prescribing profile.
Weight Loss Results with Phentermine
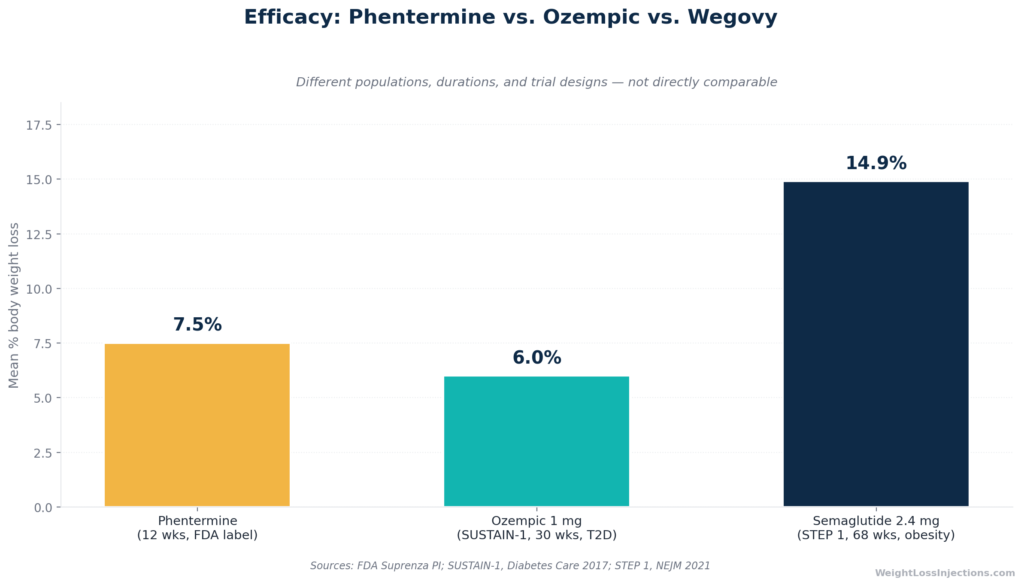
The Suprenza FDA label is notably modest in its efficacy claims, acknowledging that phentermine-treated patients in short-term trials lost “only a fraction of a pound a week” more than placebo, and that study designs make it difficult to separate drug effects from other variables. Real-world and meta-analytic estimates generally place average phentermine-induced weight loss at 5–10% of body weight over 12 weeks when combined with a reduced-calorie diet, a figure used by most clinical comparisons and the standard cited in prescriber education.
BMI eligibility per the FDA label: BMI ≥ 30 kg/m², or BMI ≥ 27 kg/m² with at least one weight-related comorbidity such as hypertension, diabetes, or hyperlipidemia.
Phentermine Side Effects (from FDA Label)
Per the DailyMed phentermine HCl prescribing information, adverse reactions include:
- Cardiovascular: Palpitation, tachycardia, elevated blood pressure, ischemic events, and — rarely — primary pulmonary hypertension or cardiac valvular disease
- Central nervous system: Insomnia, restlessness, dizziness, overstimulation, headache, euphoria, tremor
- Gastrointestinal: Dry mouth, unpleasant taste, constipation, diarrhea
- Endocrine: Decreased libido, impotence
What Is Ozempic (Semaglutide)?
Ozempic is the brand name for injectable semaglutide, FDA-approved on December 5, 2017 (NDA 209637) for glycemic control in adults with type 2 diabetes. It is not FDA-approved for weight management under the Ozempic label, that indication belongs to its sister product Wegovy (semaglutide 2.4 mg, approved June 4, 2021). However, Ozempic is widely prescribed off-label for weight loss, and the underlying molecule is identical to Wegovy.
How Ozempic Works
Semaglutide is a GLP-1 receptor agonist, a class of drugs that mimics the action of glucagon-like peptide-1, a gut hormone released after eating. Per the Ozempic FDA prescribing information, it works by stimulating insulin secretion, suppressing glucagon, slowing gastric emptying, and acting on central nervous system receptors to reduce appetite. The result is a pronounced reduction in hunger and food intake that persists as long as the medication is taken.
Unlike phentermine, which produces appetite suppression through an adrenaline-like stimulant mechanism, semaglutide works by mimicking a natural gut hormone. This distinction matters: semaglutide does not raise heart rate or blood pressure and has actually demonstrated cardiovascular protection in the SELECT trial, which led to a CV risk-reduction indication for Wegovy in March 2024.
Dosage and Duration
Ozempic is administered as a once-weekly subcutaneous injection. The titration schedule starts at 0.25 mg per week for four weeks, escalating to 0.5 mg, and potentially to 1 mg or 2 mg based on tolerability and response. This graduated approach is designed to reduce GI side effects, particularly nausea. Unlike phentermine, semaglutide is intended for long-term continuous use, the clinical trials supporting its efficacy ran 30 to 68 weeks, with evidence of continued benefit beyond two years.
Weight Loss Results with Ozempic (Semaglutide)
The key efficacy benchmarks come from two sources:
- SUSTAIN-1 trial (Ozempic doses, type 2 diabetes population, 30 weeks): Published in Diabetes Care, SUSTAIN-1 found mean body weight losses of 4.5% on semaglutide 0.5 mg and 6.0% on semaglutide 1.0 mg.
- STEP 1 trial (Wegovy dose, 2.4 mg, obesity population, 68 weeks): Published in the New England Journal of Medicine, STEP 1 found a mean body weight loss of 14.9% with semaglutide 2.4 mg vs. 2.4% on placebo. Among participants on semaglutide, 86.4% lost at least 5% of body weight, 69.1% lost at least 10%, and 50.5% lost at least 15%.
The real-world Ghusn 2022 Mayo Clinic analysis in JAMA Network Open of 175 non-diabetic patients on semaglutide found 5.9% loss at 3 months and 10.9% at 6 months, closely tracking the trial data at Ozempic doses.
Important note: Ozempic’s weight-loss use is off-label. Patients seeking the full 2.4 mg dose with a formal weight-management indication would use Wegovy. In practice, many telehealth prescribers use Ozempic off-label because its cost structure and availability have historically differed from Wegovy’s.
Ozempic Side Effects (from FDA Prescribing Information)
Per FDA-pooled placebo-controlled trial data in the Ozempic PI, the most common GI adverse events at 0.5 mg and 1 mg doses respectively were:
| Adverse event | Semaglutide 0.5 mg | Semaglutide 1 mg | Placebo |
|---|---|---|---|
| Nausea | 15.8% | 20.3% | 6.1% |
| Vomiting | 5.0% | 9.2% | 2.3% |
| Diarrhea | 8.5% | 8.8% | 1.9% |
| Constipation | 5.0% | 3.1% | 1.5% |
The FDA label also carries a boxed warning for thyroid C-cell tumors (based on rodent data), and semaglutide is contraindicated in patients with a personal or family history of medullary thyroid carcinoma or Multiple Endocrine Neoplasia syndrome type 2.
Head-to-Head Comparison
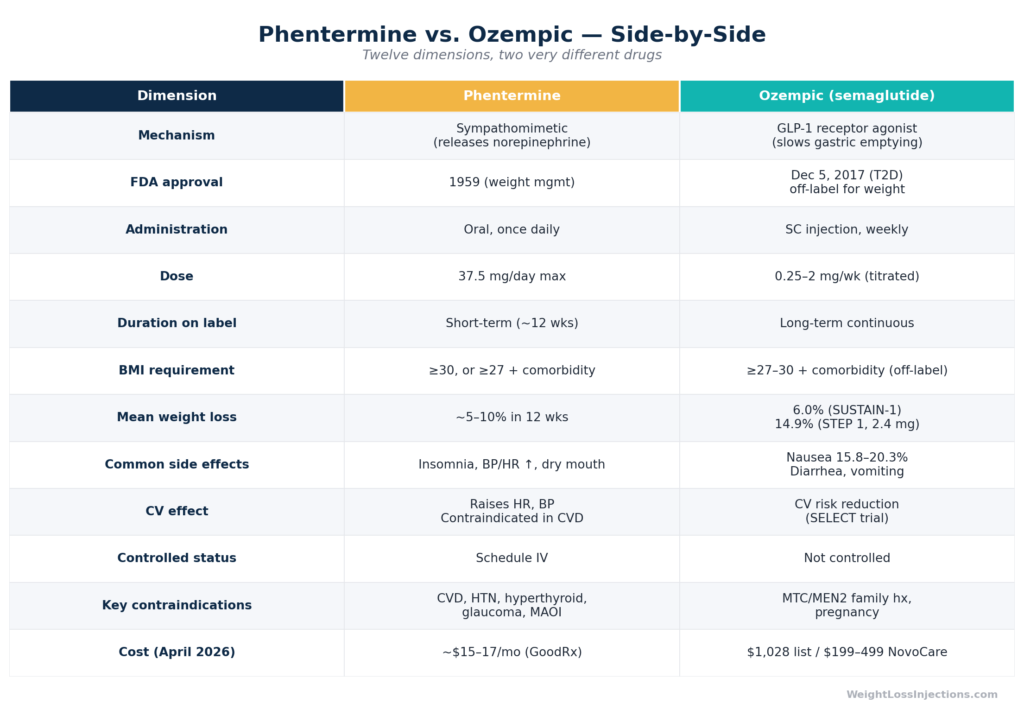
Phentermine vs. Ozempic Side-by-Side Comparison Table
Efficacy
On pure weight loss numbers, semaglutide at its full approved weight-management dose wins decisively: 14.9% loss at 68 weeks in the STEP 1 trial vs. 5–10% in roughly 12 weeks with phentermine per the standard clinical estimate. But a direct head-to-head is complicated, phentermine has not been studied in modern randomized controlled trials of comparable design and duration to the STEP program. The trials are different populations, different durations, and different eras of research methodology.
What the evidence does support: phentermine produces faster initial results (appetite suppression kicks in within hours of the first dose), while semaglutide produces greater cumulative loss over months to years. Patients who need a quick 10-pound loss for a medical procedure before a certain date are in a different situation than patients managing chronic obesity over years.
Side Effects
The adverse effect profiles of these two drugs are almost entirely non-overlapping. Phentermine’s risks are cardiovascular and stimulant-related: insomnia, elevated blood pressure, palpitations, and dry mouth dominate. Ozempic’s risks are predominantly gastrointestinal: nausea (in 15–20% of patients per FDA data), vomiting, and diarrhea that typically peak during dose escalation and improve over weeks. Patients who cannot tolerate GI effects may prefer phentermine; patients with cardiovascular disease, hyperthyroidism, or glaucoma cannot safely take phentermine at all.
Neither drug is completely benign. Phentermine’s stimulant properties have historically raised concern about habituation (it is Schedule IV), and the cardiovascular contraindications are firm. Semaglutide’s rare but serious risks, pancreatitis, gallbladder disease, thyroid C-cell tumor potential, require appropriate patient screening.
Cost
This is the starkest difference between the two drugs.
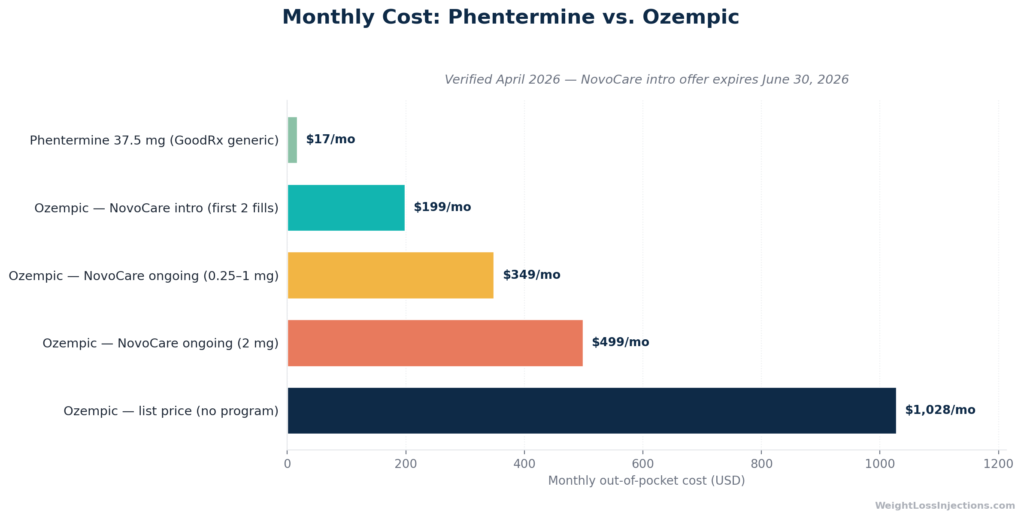
- Phentermine (generic HCl 37.5 mg): Via GoodRx, 30 tablets run approximately $16–$17 per month, making it one of the most affordable prescription weight-loss medications available.
- Ozempic: The list price is $1,028/month. Through the NovoCare Pharmacy program, patients who qualify for the self-pay program can access it at $199/month for the first two fills (0.25/0.5 mg doses, new patients only, through June 30, 2026), then $349/month (for 0.5–1 mg doses) or $499/month (for the 2 mg dose) ongoing. Without insurance or an assistance program, Ozempic is prohibitively expensive for most patients. Notably, a 2027 Novo Nordisk list-price reset announced February 2026 will bring all semaglutide forms to approximately $675/month starting in 2027.
For patients paying out-of-pocket and managing tight budgets, phentermine’s cost advantage is overwhelming, the annual cost of phentermine is roughly the same as a single month of Ozempic at list price. The relevant comparison is not always list price vs. GoodRx, however: many insured patients with type 2 diabetes have Ozempic covered at copay, while weight-only indications remain poorly covered across commercial payers.
Eligibility and Contraindications
| Category | Phentermine | Ozempic (off-label weight loss) |
|---|---|---|
| BMI requirement | ≥30, or ≥27 with comorbidity | Typically ≥27–30 with comorbidity (provider discretion) |
| Cardiovascular disease | Contraindicated | Permissible; CV-protective in SELECT trial |
| Hypertension | Contraindicated if uncontrolled | No contraindication |
| Hyperthyroidism | Contraindicated | No contraindication |
| Glaucoma | Contraindicated | No contraindication |
| Pregnancy | Contraindicated | Contraindicated; discontinue ≥2 months before planned pregnancy |
| MAOI use | Contraindicated within 14 days | No interaction |
| Thyroid cancer history (MTC/MEN2) | No restriction | Contraindicated |
| Drug abuse history | Contraindicated | No restriction |
| Controlled substance | Yes (Schedule IV — requires DEA-regulated prescribing) | No |
The practical effect: phentermine’s cardiovascular contraindications eliminate a large fraction of the obese patient population, many of whom have hypertension, coronary artery disease, or arrhythmias as comorbidities. Ozempic, by contrast, is safe — and actually beneficial — in patients with established cardiovascular disease.
Cost Comparison
Monthly Cost Comparison: Phentermine vs. Ozempic
The cost gap between phentermine and Ozempic is not just large, it is a different order of magnitude. At approximately $16–17/month via GoodRx, phentermine is accessible to almost any patient without insurance. At $1,028/month list price per verified NovoCare data, Ozempic is inaccessible without insurance coverage or assistance programs.
For patients pursuing semaglutide out-of-pocket, the NovoCare Pharmacy self-pay program is the most important pricing option to know. The introductory $199/month offer covers only the first two fills (initiation doses of 0.25 and 0.5 mg) for new patients without insurance, and it is time-limited through June 30, 2026. After that, the $349/month ongoing tier applies for 0.5–1 mg doses. Patients titrating to 2 mg face the $499/month tier. These are still more than 20 times the cost of phentermine.
One practical note: compounded semaglutide was widely available through 503A and 503B pharmacies during the FDA shortage period, sometimes at lower price points. However, the FDA declared the semaglutide shortage resolved on February 21, 2025, and the compounding deadline for 503A pharmacies passed April 22, 2025, with the 503B deadline following May 22, 2025. As of the time of this writing, compounded semaglutide from traditional compounders is no longer legally available in the standard market.
Telehealth Access at WeightLossInjections.com
Getting either medication requires a licensed prescriber evaluation, and telehealth has made that faster and more accessible than ever. At WeightLossInjections.com, the process starts with a free online eligibility intake that covers your medical history, current medications, BMI, and weight-loss goals. A licensed provider reviews your intake within 24 hours.
Our telehealth platform supports access to both options:
- Phentermine: Available to qualifying patients via telehealth in most states. Because phentermine is a Schedule IV controlled substance, prescribing is subject to DEA regulations. [service detail] Our providers can issue a controlled-substance prescription electronically in states where this is permitted, which is sent to your preferred pharmacy for same-day or next-day pickup.
- Ozempic / Semaglutide: Telehealth-eligible patients with type 2 diabetes (on-label) or qualifying obesity (off-label, at provider discretion) can receive a prescription through WeightLossInjections.com’s network. We help coordinate access to the NovoCare self-pay program and provide a personalized titration plan with ongoing provider access.
The bundled WeightLossInjections.com program is [$X/month], which includes [service detail]. Given that a single uncoordinated Ozempic prescription without support can lead to months of dose-escalation errors and unmanaged side effects, the value of structured telehealth oversight is meaningful beyond the prescription itself.
Our take at WeightLossInjections.com: Most patients asking “phentermine vs. Ozempic” are actually asking a deeper question: how much do I want to spend, and how much weight do I need to lose? Phentermine is cheap, fast, and effective in the short term, but it is not a solution for patients with cardiovascular risk factors (a very large share of the overweight and obese population), and its effects are largely time-limited. Semaglutide costs more but produces roughly double the weight loss over the long term, and for patients with type 2 diabetes it delivers cardiovascular protection as a bonus. The right answer depends on your individual profile, not on which drug is more powerful in trials. That is exactly the kind of decision a licensed provider consultation exists to make.
Which Is Right for You?
Neither medication is universally superior. Use this framework to think through your situation:
Phentermine may be more appropriate if:
- You have a short-term weight-loss goal (e.g., qualifying for a procedure, losing initial weight before a special event)
- You have a tight budget and cannot access Ozempic through insurance or assistance programs
- You have no history of cardiovascular disease, hypertension, hyperthyroidism, glaucoma, or drug abuse
- You prefer oral medication over weekly injections
- You understand the short-term-only limitation and are using it as a jump-start, not a long-term strategy
Ozempic (semaglutide) may be more appropriate if:
- You have type 2 diabetes and need both glucose control and weight management
- You have cardiovascular risk factors or established cardiovascular disease
- You are aiming for >10% body weight loss, which phentermine’s short-term window rarely achieves
- You can access it through insurance, a patient assistance program, or a telehealth bundle
- You want a long-term weight management strategy with a medication designed for sustained use
Patients who should avoid phentermine entirely: Per the DailyMed label, phentermine is contraindicated in patients with any history of cardiovascular disease, uncontrolled hypertension, hyperthyroidism, glaucoma, agitated states, history of drug abuse, pregnancy, or who are taking or have taken MAOIs within the past 14 days.
For patients who want the benefits of combination therapy, phentermine/topiramate (Qsymia) offers a lower phentermine dose alongside anticonvulsant-assisted appetite suppression and has FDA approval for chronic weight management, worth discussing with your provider if standalone phentermine is insufficient or not covered by your prescriber.
Frequently Asked Questions
Can I take phentermine and Ozempic together?
No clinical trial data supports the concurrent use of phentermine and Ozempic, and this combination is not FDA-approved or standard clinical practice. Both drugs suppress appetite through different mechanisms, and combining CNS stimulants with GLP-1 agonists raises theoretical cardiovascular concerns. Per the phentermine prescribing information, phentermine is contraindicated in patients with cardiovascular disease — a population for whom Ozempic is increasingly used. Any consideration of combined or sequential use should involve a detailed discussion with your prescribing provider about your specific cardiac and metabolic profile. Do not self-prescribe or combine these medications without explicit provider supervision.
Is Ozempic covered by insurance for weight loss?
Generally, no, at least not without a type 2 diabetes diagnosis. Most commercial insurance plans cover Ozempic for its FDA-approved indication (type 2 diabetes management) but do not cover it for off-label weight loss alone. Wegovy, which carries the formal weight-management approval, has somewhat broader coverage, but obesity medication coverage remains inconsistent across payers. The NovoCare Pharmacy self-pay program is the primary out-of-pocket access pathway, offering $199/month for the first two fills and $349/month ongoing for qualified patients. Medicare Part D covers Wegovy for cardiovascular risk reduction (following the SELECT trial indication added in March 2024) for qualifying beneficiaries, a separate pathway worth exploring with your insurance plan.
How fast does phentermine work vs. Ozempic?
Phentermine’s appetite suppression begins within hours of the first dose, most patients notice reduced hunger on day one. Weight loss typically starts in the first week as caloric intake drops. Ozempic, by contrast, works gradually. The 0.25 mg starting dose is a non-therapeutic initiation dose per the Ozempic FDA label, meaning meaningful appetite suppression generally begins around weeks 4–8, after reaching the 0.5–1 mg maintenance range. Steady-state plasma levels are not reached for 4–5 weeks from initiation. This matters for patient expectations: phentermine users often see their fastest results in week one or two, while Ozempic users typically see their steepest loss rate in months two through six. The STEP 1 trial showed progressive weight loss continuing all the way to week 68, a trajectory impossible with short-term phentermine.
What is the BMI requirement for these medications?
Both medications use similar BMI thresholds per their respective FDA labels. For phentermine, per the Suprenza FDA prescribing information: initial BMI ≥ 30 kg/m², or ≥ 27 kg/m² with at least one weight-related risk factor (controlled hypertension, diabetes, hyperlipidemia). The Ozempic FDA label is for type 2 diabetes and does not specify a BMI cutoff for that indication. For off-label weight-loss use of Ozempic, most providers apply the Wegovy criteria: BMI ≥ 30 kg/m², or ≥ 27 kg/m² with at least one weight-related comorbidity. In practice, telehealth providers assess individual patient profiles holistically, BMI is one input, not the only gatekeeping criterion.
How do phentermine and Ozempic side effects compare?
The two drugs have almost entirely distinct side effect profiles. Phentermine, per the DailyMed label, primarily causes cardiovascular and CNS stimulant effects: insomnia, restlessness, elevated blood pressure, palpitations, tachycardia, and dry mouth. These reflect its mechanism as a norepinephrine-releasing sympathomimetic. Ozempic, per FDA pooled clinical trial data, primarily causes GI effects: nausea (15.8–20.3% vs. 6.1% placebo), vomiting (5.0–9.2% vs. 2.3%), and diarrhea (8.5–8.8% vs. 1.9%). Ozempic’s GI side effects are most pronounced during dose escalation and typically improve with time. Phentermine’s cardiovascular effects persist as long as the drug is taken. For patients who cannot tolerate GI symptoms, phentermine may be subjectively more comfortable. For patients with cardiac risk, phentermine’s side effect profile may be medically unacceptable.
This article is for educational purposes only and does not constitute medical advice. WeightLossInjections.com’s editorial team reviews all content quarterly; last medical review: April 2026. Weight loss results vary by individual; clinical trial averages represent population means, not guaranteed outcomes. Consult a licensed provider before starting any weight-loss medication.
Find out which medication is right for you. Take our free online eligibility intake — it takes under 3 minutes. A licensed WeightLossInjections.com provider reviews your history within 24 hours and can help you determine whether phentermine, Ozempic, or another option is appropriate for your situation.
Start your free intake →

