Ozempic Access Path Cost Comparison
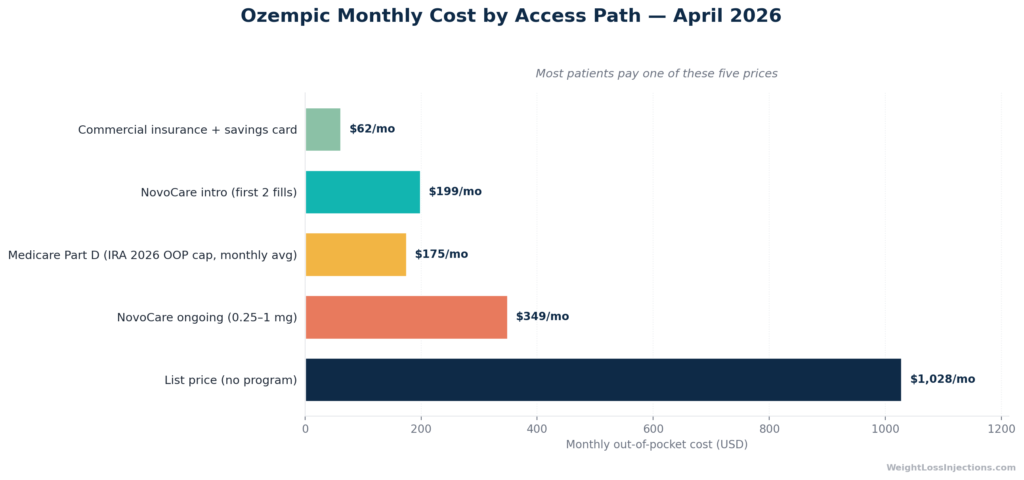
Ozempic’s official list price is $1,028/mo, but that is rarely what anyone pays. Commercial insurance with a Novo Nordisk savings card brings it to as little as $25/mo. The NovoCare direct self-pay program offers $199/mo for the first two fills (new patients, through June 30, 2026), then $349/mo ongoing for 0.25–1 mg doses. Medicare Part D enrollees face a $2,100 annual out-of-pocket cap in 2026 under the Inflation Reduction Act.
Compounded semaglutide is no longer legally permissible for most patients following the FDA’s May 2025 enforcement deadlines. A 35% list-price cut to $675/mo for all Novo semaglutide forms takes effect January 1, 2027.
Why Ozempic Is So Expensive: The List Price vs. Net Price Gap
To understand Ozempic’s price, you first need to understand that the $1,028/mo list price is largely a fiction, a sticker number that almost nobody pays. The reality of U.S. drug pricing is a negotiated system between Novo Nordisk, pharmacy benefit managers (PBMs), insurers, and pharmacies, with rebates flowing behind the scenes that can reduce what the system actually nets for the manufacturer by 40% to 70%.
How PBM Rebates Drive Up the List Price
PBMs, companies like CVS Caremark, Express Scripts, and OptumRx, negotiate formulary placement for insurers. To earn preferred tier status (meaning your drug is covered at a low copay), manufacturers pay rebates to PBMs. Those rebates are calculated as a percentage of the list price. This structure creates a perverse incentive: a higher list price enables a larger rebate, which buys better formulary position, which drives more volume. The result is that Novo Nordisk’s list price for Ozempic is set high in part to fund the rebate machinery that keeps it on commercial formularies.
What the manufacturer actually receives, the net price, is substantially lower than the list price. For Ozempic, the gap between list and net has been estimated in industry analyses as potentially exceeding 50% for commercially insured patients, per IQVIA analysis of U.S. pharmaceutical pricing dynamics. That means the $1,028 list price is, in practical terms, an accounting construct that primarily affects patients who fall outside the insurance and assistance system, uninsured individuals paying cash at retail.
The Patent Cliff: Why Generic Semaglutide Is Still Years Away
The other structural driver of Ozempic’s cost is patent protection. Novo Nordisk holds a layered portfolio of U.S. patents on semaglutide that extends well past the drug’s 2017 approval. The most commercially significant are the acylated GLP-1 compound patents (US8129343), which run through December 5, 2031, and the long-acting GLP-1 peptide use patents (US10335462) expiring June 21, 2033. Additional formulation and device patents extend protection into the late 2030s.
This means that while semaglutide’s compound patents have begun expiring in markets like India, China, Brazil, and Canada, enabling generic competition abroad, U.S. semaglutide exclusivity runs through at least 2031–2032, per patent analysis by Chemical & Engineering News. Patients in the United States will not see a generic equivalent of Ozempic at their pharmacy before the early 2030s under current projections.
The practical implication: there is no generic Ozempic coming soon. The cost paths that exist now, insurance, savings programs, and telehealth bundles, are the cost paths for the foreseeable future.
The 2026 Price Landscape: Every Access Path in One Place
GLP-1 Cash Price Comparison Table 2026
Ozempic List Price: $1,028/mo
The Ozempic list price as of April 2026 is $1,028/mo, per GoodRx and confirmed by Novo Nordisk’s own pricing materials. This is the price paid by anyone without insurance coverage, without a savings card, and outside any manufacturer assistance program. It applies to all three Ozempic pen configurations (red/blue/yellow, covering 0.25 mg through 2 mg doses). In practice, this price is primarily relevant for uninsured patients who are unaware of, or ineligible for, Novo Nordisk’s self-pay programs.
NovoCare Self-Pay Program (Best Cash Option for Most Patients)
Novo Nordisk’s NovoCare Pharmacy self-pay program is the single most important pricing mechanism for uninsured or self-paying Ozempic patients. The tiers as of April 2026:
| Program Tier | Dose | Monthly Cost | Notes |
|---|---|---|---|
| Introductory offer (new patients only) | 0.25 mg or 0.5 mg | $199/mo | First 2 fills only; offer available through June 30, 2026 |
| Standard ongoing self-pay | 0.25, 0.5, or 1 mg | $349/mo | After intro period |
| Standard ongoing self-pay | 2 mg | $499/mo | No intro discount at this dose |
Source: NovoCare Pharmacy savings page; Novo Nordisk Nov. 2025 press release; verified April 2026.
Eligibility nuances: Government beneficiaries, Medicare, Medicaid, TRICARE, VA, are explicitly excluded from the NovoCare self-pay pricing and from the commercial savings card. The program runs outside of insurance and does not count toward deductibles or out-of-pocket maximums. The introductory $199/mo offer is for patients new to both the Ozempic Savings Offer and NovoCare Pharmacy; existing savings card users who transition to NovoCare are eligible if they have not previously used the NovoCare channel.
Commercial Insurance: As Low as $25/mo
Commercially insured patients whose plans cover Ozempic (typically for type 2 diabetes) can use Novo Nordisk’s insurance savings card to pay as little as $25/mo, subject to a maximum savings of $100/month. The offer is valid for up to 48 months. In real-world terms, most commercially insured patients on a preferred formulary tier pay between $25 and $100/mo out of pocket, depending on their plan’s cost-sharing structure.
Off-label weight loss coverage is the critical asterisk. Ozempic is FDA-approved for type 2 diabetes, not weight loss. Weight loss is an off-label use. Most commercial insurance plans do not cover Ozempic for weight loss as a standalone indication, they cover Wegovy (the FDA-approved weight management formulation at 2.4 mg) for obesity management, subject to prior authorization. Patients prescribed Ozempic specifically for weight management should expect coverage denial and plan accordingly. The $25 copay path almost exclusively applies to patients with a type 2 diabetes diagnosis on their plan.
Medicare Part D 2026: $2,100 Annual OOP Cap
For Medicare beneficiaries, the Inflation Reduction Act of 2022 established a hard annual out-of-pocket cap for Medicare Part D covered medications. The cap is $2,100 in 2026, per GoodRx Medicare Part D OOP maximum analysis. After spending $2,100 on covered drugs in a calendar year, the plan pays 100% of costs for the remainder of the year.
For Ozempic as a diabetes medication, this translates to a maximum OOP exposure of $2,100/year, roughly $175/month on average, assuming coverage all year. After that threshold, the drug effectively costs $0 for the rest of the year. However, Medicare coverage for Ozempic specifically for weight loss remains limited; beneficiaries should verify their plan’s formulary status with their Part D carrier before assuming coverage.
Separately, CMS negotiated a 71% discount on Novo Nordisk’s semaglutide products under the IRA Medicare price negotiation program. That negotiated price of approximately $274/month for Ozempic, Wegovy, and Rybelsus takes effect January 1, 2027, for Medicare patients.
GoodRx and Pharmacy Coupon Prices
GoodRx currently shows Ozempic prices starting at $199 at many pharmacies, effectively reflecting the NovoCare self-pay pricing that flows through the GoodRx platform. Retail pharmacy prices without any discount card or program vary by location, typically $850–$1,100 depending on chain and region. Patients using GoodRx or SingleCare should verify ZIP-code-specific pricing at their pharmacy before filling; prices differ by a meaningful margin between chains and metro areas.
Cash Price Without Insurance: Total Monthly Cost for Weight Loss Patients
The medication cost is one piece of a larger picture for Ozempic weight loss patients. The actual monthly cost of a supervised Ozempic program includes:
- Medication: $349/mo (NovoCare ongoing, 0.25–1 mg) to $499/mo (2 mg)
- Telehealth consultation: Typically $100–$200/mo depending on the provider and whether visits are bundled
- Labs: GLP-1 monitoring labs (metabolic panel, HbA1c, lipids) typically run $50–$150 per draw; frequency varies by provider protocol
12-month cost example at NovoCare $349/mo:
- Medication: 12 × $349 = $4,188
- Telehealth visits (bundled quarterly check-ins, estimate): $400–$600
- Labs (2–3 draws per year): $150–$300
- Estimated annual total: $4,738–$5,088
6-month example at NovoCare introductory rates ($199 × 2, then $349 × 4):
- Medication: ($199 × 2) + ($349 × 4) = $1,794
- Telehealth + labs (6 months): $250–$400
- Estimated 6-month total: $2,044–$2,194
These are ballpark figures. Telehealth providers with bundled models can significantly reduce the non-medication overhead. At WeightLossInjections.com, our bundled program starts at [$X/month] and includes [service detail].
Insurance Coverage for Ozempic Weight Loss: The Prior Auth Reality
Attempting to get commercial insurance to cover Ozempic for off-label weight loss requires navigating a narrow and often frustrating pathway.
Why Plans Deny Off-Label Coverage
Ozempic’s FDA-approved indications are type 2 diabetes glycemic control (since December 5, 2017, NDA 209637), cardiovascular risk reduction, and chronic kidney disease. Weight management is not among them. Most commercial plans follow FDA indications in their coverage policies, directing weight-loss patients toward Wegovy (FDA-approved for obesity) rather than Ozempic. The practical result is that plans covering Wegovy for weight management at $25/mo after a savings card will decline the same semaglutide molecule when prescribed as Ozempic for the same purpose.
Step Therapy and Prior Authorization
Even for patients with a legitimate type 2 diabetes diagnosis, most commercial plans require:
- Prior authorization confirming the diagnosis and clinical appropriateness
- Step therapy — documentation that you have tried and failed at least one other diabetes medication (often metformin) before escalating to a GLP-1 agonist
- BMI and comorbidity criteria for Wegovy-specific weight management coverage, typically BMI ≥30 or BMI ≥27 with at least one weight-related condition
Providers can submit appeal letters documenting the clinical rationale for specific cases. The Ozempic FDA prescribing information §1 supports cardiovascular and glycemic benefit in relevant populations, which forms the basis of most successful appeals for patients with comorbid diabetes and obesity.
The End of Compounded Semaglutide: What Happened and Why It Matters
For two years — roughly 2022 through early 2025 — compounded semaglutide was widely available through telehealth companies as a cheaper alternative to branded Ozempic and Wegovy. That window is now legally closed for the vast majority of patients, and patients still being offered “compounded semaglutide” at a discount should understand the current regulatory reality.
The Timeline
- 2022–early 2025: FDA listed semaglutide on its official drug shortage list. Under federal law (21 U.S.C. § 503A and § 503B), shortage status authorized compounding pharmacies and outsourcing facilities to compound copies of shortage-listed drugs.
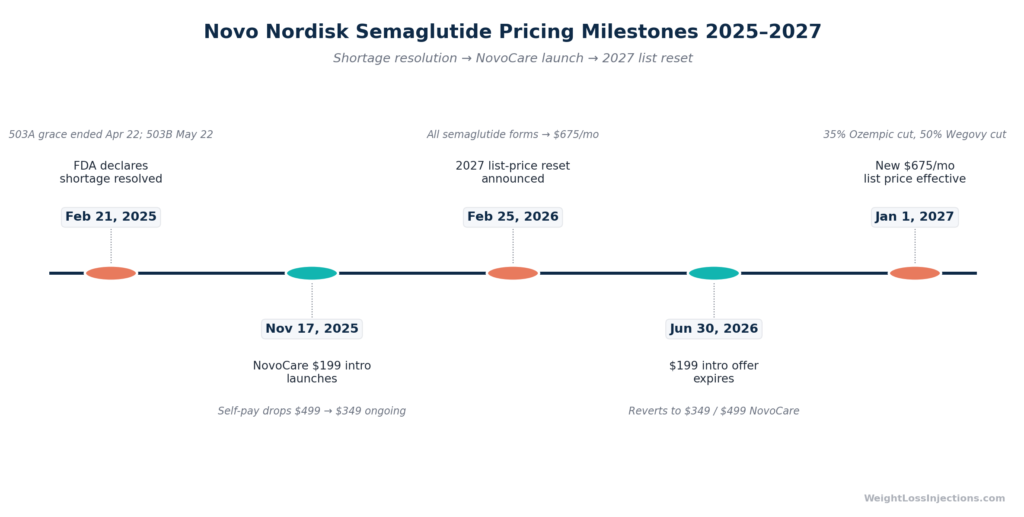
- February 21, 2025: FDA declared the semaglutide shortage resolved, per the FDA drug shortage resolution order. Shortage-status authorization to compound immediately began phasing out.
- April 22, 2025: Enforcement deadline for 503A (patient-specific compounding pharmacies). After this date, compounding semaglutide as an essentially copy of an FDA-approved product is no longer permitted, per FDA 503A compounding guidance.
- May 22, 2025: Enforcement deadline for 503B (outsourcing facilities handling bulk supply). After this date, large-scale compounded semaglutide production and distribution became prohibited, per FDA outsourcing facility guidance and Alston & Bird legal analysis (March 2025).
What Narrow Exceptions Still Exist
503A pharmacies can still compound semaglutide for individual patients when a prescriber documents a specific clinical need not met by the approved product, for example, a medically necessary dose not commercially available, or a documented allergy to an inactive ingredient. But this is a documented, patient-specific exception, not a general population offering. Per legal analysis from Nixon Law Group, any telehealth platform offering compounded semaglutide without documenting a patient-specific medical need is operating outside FDA guidance.
Our take at WeightLossInjections.com: If a telehealth provider is currently marketing “compounded semaglutide” or “compounded Ozempic” as a general cash-pay alternative, months after the May 2025 enforcement deadline, that product is not legally authorized under FDA’s shortage-exemption framework. The legitimate self-pay paths are Novo Nordisk’s NovoCare program or GoodRx-priced branded Ozempic at a retail pharmacy. WeightLossInjections.com uses only FDA-approved, branded medications. No compounded products. No regulatory shortcuts.
GLP-1 Price Comparison: Where Ozempic Fits in the 2026 Market
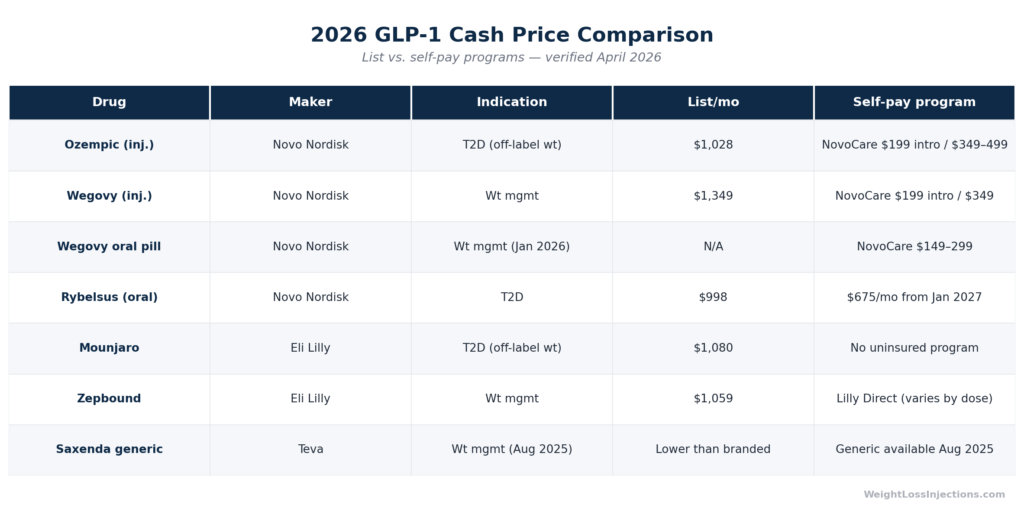
Ozempic does not exist in a vacuum. For weight-loss patients evaluating their options, the relevant comparison set is the full GLP-1 landscape:
| Drug | Active Ingredient | Weight Loss Approval | 2026 List Price | Best Cash Path |
|---|---|---|---|---|
| Ozempic | Semaglutide inj. | Off-label only | $1,028/mo | NovoCare: $199 intro / $349 ongoing |
| Wegovy | Semaglutide inj. 2.4 mg | FDA-approved (obesity) | $1,349/mo | NovoCare: $199 intro / $349 ongoing |
| Wegovy oral pill | Oral semaglutide | FDA-approved (obesity) | Launched Jan 2026 | NovoCare: $149–$299/mo |
| Rybelsus | Oral semaglutide | Off-label only (diabetes) | $997.58/mo | No uninsured program; $675/mo from Jan 2027 |
| Mounjaro | Tirzepatide inj. | Off-label only | ~$1,080/mo | No uninsured program (commercial savings card only) |
| Zepbound | Tirzepatide inj. | FDA-approved (obesity) | ~$1,059/mo | Eli Lilly Direct: varies by dose |
| Saxenda generic | Liraglutide (Teva) | FDA-approved (obesity) | Lower than branded | Available August 28, 2025 |
Sources: SHARED_SPEC verified facts April 2026; NovoCare Pharmacy; CNN Novo Nordisk pricing coverage (Feb 2026); GoodRx Ozempic.
Wegovy vs. Ozempic pricing note: For weight management specifically, Wegovy has FDA approval at 2.4 mg weekly, a dose not available with Ozempic’s labeled indications. The STEP 1 trial published in NEJM demonstrated a mean 14.9% body weight loss at 68 weeks with semaglutide 2.4 mg (Wegovy). Ozempic’s weight-loss data in a type 2 diabetes population from SUSTAIN-1 showed 4.5% loss at 0.5 mg and 6.0% at 1.0 mg at 30 weeks, a meaningfully lower effect, in a different population, at lower doses. Per the real-world Mayo Clinic study in JAMA Network Open (Ghusn 2022), average weight loss on semaglutide was 5.9% at 3 months.
Mounjaro (tirzepatide) is worth a separate mention: despite its ~$1,080/mo list price, there is no direct uninsured/self-pay program equivalent to NovoCare. Cash-pay patients without commercial insurance have no manufacturer-supported discount path for Mounjaro, making its effective cash cost the full list price.
The 2027 List Price Reset: What Changes in January
Novo Nordisk 2027 Semaglutide Price Timeline
On February 25, 2026, Novo Nordisk announced that all forms of semaglutide, injectable Ozempic, injectable Wegovy, oral Wegovy, and Rybelsus, will be reset to a $675/month list price effective January 1, 2027. For Ozempic (current list $1,028/mo), this represents a 34% reduction. For Wegovy (current list $1,349/mo), the cut is approximately 50%.
Per CNN coverage of the announcement and GastroenterologyAdvisor, the $675/mo list price is designed to benefit patients whose costs are directly tied to the list price, primarily uninsured cash-pay patients not enrolled in NovoCare or GoodRx programs, and patients in certain Medicare scenarios. Patients currently enrolled in the NovoCare $349/mo or $199/mo programs will not see a direct price reduction from the 2027 list-price change, per the company’s statement that “adjustments to list prices will not affect self-pay rates.”
The relevant comparison for planning purposes:
| Scenario | 2026 Cost | Post-Jan 2027 |
|---|---|---|
| NovoCare $349/mo (ongoing) | $349/mo | Unchanged |
| NovoCare $199/mo intro | $199/mo (through June 2026) | Intro expires; $349/mo resumes |
| List price, no program | $1,028/mo | $675/mo |
| Medicare Part D (CMS negotiated) | Varies (OOP cap $2,100/yr) | ~$274/mo effective Jan 2027 |
The list-price cut matters most for the estimated segment of patients paying full retail — a relatively small but financially burdened group. For patients who are already on NovoCare, the practical change is minimal until January 2027 brings the negotiated Medicare prices into effect.
Is Ozempic Worth the Cost for Weight Loss?
The clinical case for Ozempic’s off-label use in weight management is solid but dose-dependent. Ozempic is approved at doses up to 2 mg for diabetes management. In the weight-loss context, meaningful results require extended treatment, and the efficacy data comes from different dose ranges.
What the Data Actually Shows
The STEP 1 trial (New England Journal of Medicine, 2021), which studied semaglutide at 2.4 mg weekly (Wegovy’s dose), showed a mean 14.9% body weight loss at 68 weeks compared to 2.4% with placebo in adults without diabetes. That is the benchmark for maximum semaglutide efficacy at an approved weight-loss dose.
At Ozempic’s licensed doses for diabetes, the SUSTAIN-1 trial showed 4.5% body weight loss at 0.5 mg and 6.0% at 1.0 mg at 30 weeks in a type 2 diabetes population. Real-world data from Ghusn et al. 2022 in JAMA Network Open (Mayo Clinic) showed 5.9% weight loss at 3 months in a mixed-indication semaglutide population.
Cost-Per-Result Framing
Cost Per Pound Lost: Ozempic NovoCare Pricing Analysis
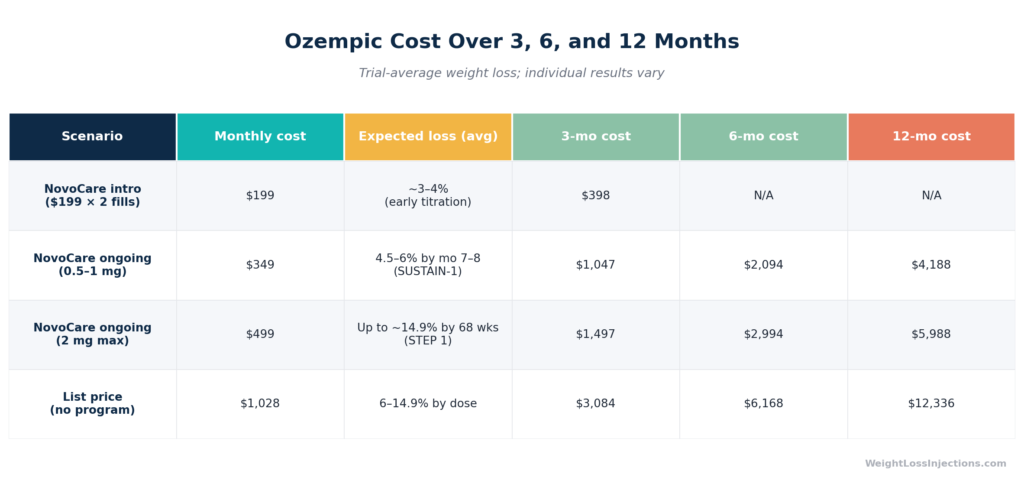
At $349/mo via NovoCare, twelve months of Ozempic costs $4,188 in medication alone. For a 200-pound person achieving the SUSTAIN-1 average of 6% weight loss at 1 mg, that equates to roughly 12 pounds, approximately $349 per pound lost, excluding telehealth and lab costs. At the higher 14.9% STEP 1 efficacy (Wegovy dosing), the same person loses 30 pounds at $140/lb over 68 weeks.
These figures are averages from controlled trials. Individual results vary substantially. The STEP 1 trial showed a range of responses, roughly 31% of participants lost more than 20% of body weight, while a meaningful proportion lost less than 5%. Drug response is not uniform, which is why clinical supervision matters: non-responders should be identified early to avoid prolonged cost exposure.
The appropriate framing is not “is this drug worth $349/mo” in the abstract, it is whether supervised GLP-1 therapy, at available access prices, is cost-competitive with alternatives like bariatric surgery or long-term diet programs when valued against the documented metabolic and cardiovascular outcomes. For many patients with comorbid type 2 diabetes or cardiovascular risk, the SUSTAIN 6 cardiovascular trial (NEJM, 2016) showed a 26% relative risk reduction in MACE outcomes, a clinical benefit with calculable economic value beyond the scale.
Telehealth Access: How WeightLossInjections.com Compares
The telehealth landscape for GLP-1 medications has consolidated significantly since the compounded semaglutide window closed. Providers now operate in one of two models: they help patients access branded Ozempic (or Wegovy) through NovoCare-compatible prescriptions, or they bundle the prescription and clinical management into a monthly fee.
At WeightLossInjections.com, our supervised GLP-1 program provides access to FDA-approved Ozempic starting at [$X/month], which includes [service detail]. Our licensed providers handle the prescription, prior authorization coordination where applicable, and ongoing clinical check-ins, a meaningful advantage over self-navigating the NovoCare enrollment and pharmacy fulfillment process.
Comparison platforms like Ro and others have published self-pay pricing for Ozempic bundles. These typically start at a medication pass-through cost aligned with NovoCare pricing plus a telehealth management fee. The differentiator is the quality of clinical oversight, the speed of intake, and whether the provider actively assists with insurance prior authorization for eligible patients.
For patients with commercial insurance, the most valuable service a telehealth provider can offer is prior authorization support — the process of obtaining coverage approval can reduce the monthly cost from $349+ to $25. At WeightLossInjections.com, our clinical team assists with PA submissions as part of [service detail].
Our take at WeightLossInjections.com: The Ozempic pricing landscape in 2026 is more navigable than it appears from the $1,028 list price. The NovoCare $199 introductory offer is a genuine entry point for new self-pay patients. The $349/mo ongoing rate is real and accessible without income verification for most patients. What the pricing structure does not offer is clinical management, a prescription and a pen do not by themselves constitute a supervised weight-loss program. The outcomes data from STEP 1 and SUSTAIN-1 were generated in tightly managed clinical settings. Patients who get Ozempic via telehealth and then go without follow-up are not replicating those conditions. Our program starts at [$X/month] and ensures you have a licensed provider available throughout your treatment, not just at prescription initiation.
Frequently Asked Questions
How much is Ozempic for weight loss without insurance in 2026?
Without insurance, the most accessible pricing is through Novo Nordisk’s NovoCare Pharmacy self-pay program. New patients pay $199/mo for the first two fills (0.25 mg or 0.5 mg doses only; offer runs through June 30, 2026). After that, ongoing pricing is $349/mo for 0.25–1 mg doses and $499/mo for the 2 mg dose. The official list price without any program is $1,028/mo, per GoodRx Ozempic pricing — but paying that amount without first checking NovoCare or GoodRx is effectively leaving several hundred dollars on the table each month.
Does insurance cover Ozempic for weight loss?
Rarely. Ozempic is FDA-approved for type 2 diabetes, not weight loss. Most commercial plans follow FDA indications in their coverage policies, meaning they cover Wegovy (the FDA-approved weight management semaglutide formulation at 2.4 mg) rather than Ozempic for obesity. Patients with a comorbid type 2 diabetes diagnosis may access Ozempic under their diabetes coverage, though prior authorization and step therapy are commonly required. Off-label weight loss coverage for Ozempic as a standalone indication is not standard at most commercial plans. Government beneficiaries (Medicare, Medicaid) are excluded from Novo Nordisk’s savings card programs, per NovoCare eligibility terms.
What is the cheapest way to get Ozempic on a cash-pay basis?
The cheapest legal path for most uninsured patients in 2026 is the NovoCare introductory offer: $199/mo for the first two fills of 0.25 mg or 0.5 mg through June 30, 2026, per the Novo Nordisk November 2025 self-pay announcement. After that, the $349/mo ongoing rate is the best widely accessible price. GoodRx currently reflects these NovoCare-aligned prices at major pharmacy chains. Novo Nordisk’s Patient Assistance Program provides Ozempic free of charge to qualifying uninsured or underinsured patients with household income at or below 400% of the Federal Poverty Level (approximately $62,400 for an individual in 2026), per NovoCare Patient Assistance Program information. Compounded semaglutide is no longer legally authorized for general dispensing following the May 2025 FDA enforcement deadlines.
How does Ozempic’s price compare to Wegovy?
Wegovy’s list price is $1,349/mo versus Ozempic’s $1,028/mo — a $321/mo list-price premium for Wegovy. Through NovoCare, both medications share the same self-pay tiers: $199/mo introductory and $349/mo ongoing (through June 2026 for the intro offer). Clinically, Wegovy is FDA-approved for weight management at 2.4 mg, the dose that produced 14.9% average weight loss in STEP 1 (NEJM, 2021). Ozempic’s maximum labeled dose is 2 mg; off-label weight loss data at lower doses shows 4.5–6.0% loss per SUSTAIN-1. For weight loss specifically, Wegovy’s FDA-approved status also means commercial plans may cover it for obesity (with prior authorization), which is generally not available for Ozempic. Additionally, Novo Nordisk launched an oral Wegovy pill in January 2026 at $149–$299/mo through NovoCare, providing another semaglutide option at lower cost.
What will Ozempic cost in 2027?
Novo Nordisk announced on February 25, 2026, that Ozempic’s list price will be cut from $1,028/mo to approximately $675/mo effective January 1, 2027, per U.S. News & World Report and GastroenterologyAdvisor. The same $675/mo list price will apply to Wegovy, oral Wegovy, and Rybelsus. NovoCare self-pay rates ($349/mo, $499/mo) are separate from the list price and are not directly changed by the 2027 announcement. For Medicare beneficiaries, CMS-negotiated pricing of approximately $274/mo for semaglutide products also takes effect January 1, 2027. Patients currently on NovoCare programs should evaluate whether switching to insurance-covered access post-2027 makes financial sense for their situation.
Can I still get compounded semaglutide in 2026?
No, for most patients. The FDA declared the semaglutide shortage resolved on February 21, 2025, which ended the legal basis for widespread semaglutide compounding. Enforcement deadlines passed for 503A pharmacies (April 22, 2025) and 503B outsourcing facilities (May 22, 2025), per Alston & Bird legal analysis. A narrow exception exists under 503A for documented patient-specific clinical needs not met by the approved product, but general dispensing of compounded semaglutide as a cheaper alternative to branded Ozempic or Wegovy is not legally authorized. Providers still marketing “compounded semaglutide” to general patient populations in 2026 are not operating within FDA-sanctioned boundaries, per Nixon Law Group analysis.
This article is for informational purposes only and does not constitute medical advice. All pricing reflects verified sources as of April 2026; prices and program availability are subject to change. WeightLossInjections.com editorial team reviews content quarterly; last verified April 2026. Consult a licensed provider before starting or changing any medication.

