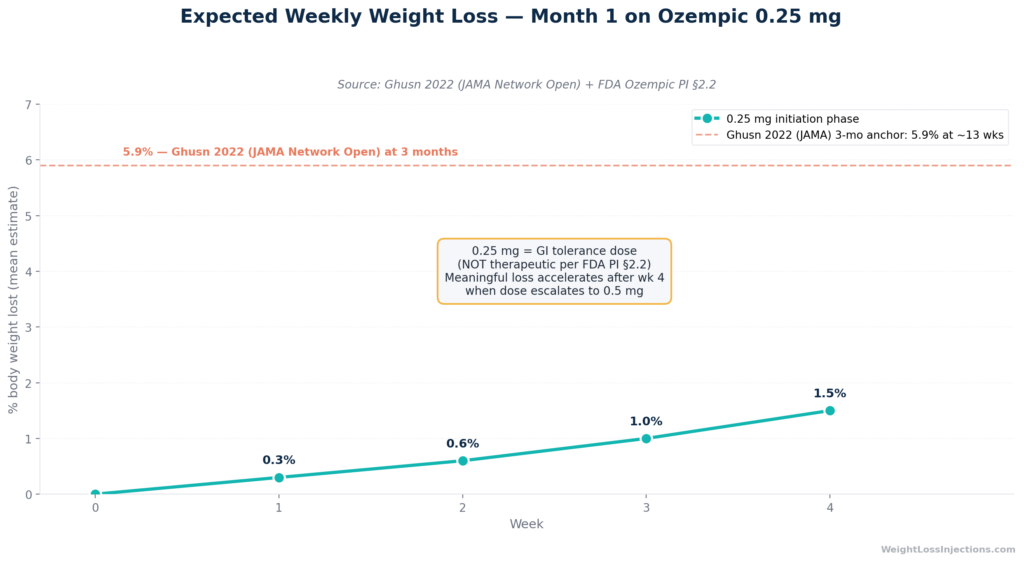
Expected Weekly % Body Weight Loss: Weeks 1–4 on Ozempic 0.25 mg
Month 1 on Ozempic is the initiation phase, not peak weight loss. You are on 0.25 mg, a dose FDA prescribing information §2.2 explicitly classifies as “not effective for glycemic control.” Most people lose 1–5 lbs in the first four weeks, and that is appropriate.
The Ghusn 2022 Mayo Clinic real-world study in JAMA Network Open tracked 175 adults on semaglutide and found a mean 5.9% body weight loss at three months, not one. The bulk of your weight loss comes after month 1.
What Ozempic Actually Does for Weight Loss
Ozempic (semaglutide injection) is FDA-approved for type 2 diabetes (NDA 209637, approved December 5, 2017), but its primary mechanism has made it a widely prescribed off-label option for weight management. Semaglutide is a GLP-1 receptor agonist, it mimics glucagon-like peptide-1, a gut hormone naturally released after eating. According to the Ozempic FDA prescribing information, the drug stimulates insulin secretion in a glucose-dependent manner, suppresses glucagon, and, critically for weight, significantly slows gastric emptying and acts on hypothalamic satiety centers in the brain to reduce appetite.
The practical effect is a sustained reduction in caloric intake without the acute hunger signals that typically make dieting difficult. Patients consistently report smaller portions, reduced cravings for calorie-dense foods, and early satiety. But this mechanism takes time to build to its full effect, and the titration schedule that governs month 1 is specifically designed to let the GI tract adapt, not to drive weight loss from day one.
Off-label status: Ozempic is not FDA-approved for weight loss as a primary indication. Wegovy (semaglutide 2.4 mg) carries that approval (NDA 215256, June 4, 2021). Physicians may legally prescribe Ozempic off-label for weight management, and many do, but patients should understand they are using a diabetes medication in a non-approved capacity.
Ozempic Dosing: Why Month 1 Is the Tolerance Phase, Not the Loss Phase
The 0.25 mg Initiation Dose: Designed for GI Adaptation, Not Therapy
The FDA-labeled Ozempic titration begins at 0.25 mg once weekly for four weeks. The purpose of this dose is unambiguous in the label: per Ozempic FDA prescribing information §2.2, the 0.25 mg starting dose is “intended for treatment initiation and is not effective for glycemic control.” It exists to allow the gastrointestinal tract to adapt to semaglutide’s gastric-emptying effect before the therapeutic dose begins, not to produce weight loss or lower blood sugar.
This has a direct implication for month 1 expectations: you are spending four weeks at a sub-therapeutic dose. Weight loss during this period, while possible, will be modest for most patients. The drug is not yet at the dose where it substantially suppresses appetite or produces the caloric deficit that drives meaningful fat loss.
After four weeks at 0.25 mg, the dose escalates to 0.5 mg weekly — the first therapeutic level. If the 0.5 mg dose does not achieve adequate clinical response after a minimum of four additional weeks, the provider may escalate to 1 mg, and then optionally to 2 mg (the maximum FDA-labeled dose), each step requiring at least four weeks to assess tolerance before escalating further, per FDA PI §2.2.
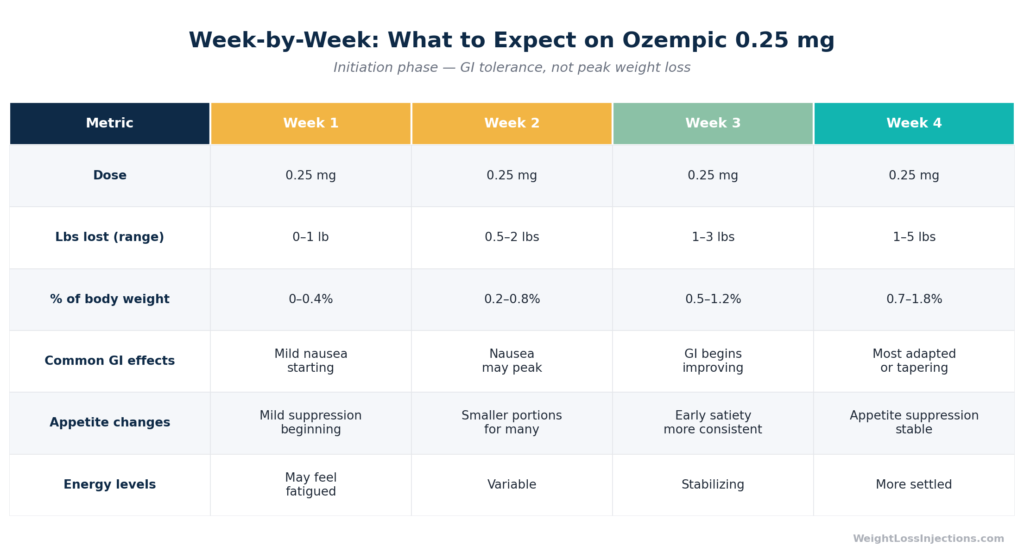
Week-by-Week Comparison Table: Month 1 on Ozempic 0.25 mg
What Month 1 Actually Costs You in Dose Time
For context: a patient who starts Ozempic on January 1 and follows the standard titration is at 0.25 mg for all of January. They do not reach the first therapeutic dose (0.5 mg) until February, and do not reach 1 mg until March at earliest. Month 1 is genuinely the slow ramp. Patients who experience this and conclude the drug “isn’t working” are making a judgment before the dose has reached a therapeutic level.
This is not a reason to skip the titration, it is a reason to reframe expectations before starting.
What Clinical Data Shows About Weight Loss in the First Month
The Ghusn 2022 Mayo Clinic Cohort: The Best Real-World Anchor
The best real-world estimate for early semaglutide weight loss comes from Ghusn et al. 2022, published in JAMA Network Open, a Mayo Clinic Health System dataset of 175 adults with overweight or obesity (mean BMI 41.3) who were prescribed subcutaneous semaglutide. This cohort reflects the closest real-world approximation of what Ozempic users experience.
Key findings:
- At 3 months: Mean body weight loss of 5.9% (6.7 kg). More than half of patients (53.7%) reached ≥5% loss; only 14.9% reached ≥10%.
- At 6 months (n=102): Mean body weight loss of 10.9% (12.3 kg), with 87.3% achieving ≥5%.
There is no month-1 data point in the Ghusn cohort because the study’s first measurement was at three months. But the math is instructive: if the 3-month mean is 5.9%, and weight loss accelerates as doses increase, a reasonable estimate for month 1 (at the 0.25 mg initiation dose) is roughly 1–2% of starting body weight, or 1–5 lbs for a person weighing 150–250 lbs.
What this means for a person starting Ozempic at different weights:
| Starting Weight | 1% Loss | 2% Loss | 5.9% (3-month avg) |
|---|---|---|---|
| 150 lbs | 1.5 lbs | 3 lbs | 8.9 lbs |
| 200 lbs | 2 lbs | 4 lbs | 11.8 lbs |
| 250 lbs | 2.5 lbs | 5 lbs | 14.8 lbs |
Month-1 estimates based on linearization from Ghusn 2022 JAMA Network Open 3-month anchor; 3-month figures reflect the actual study data.
The STEP 1 Trial: Trajectory at Therapeutic Doses
The STEP 1 trial, published in the New England Journal of Medicine, tested semaglutide 2.4 mg (Wegovy, not standard Ozempic doses) in 1,961 adults with obesity. The trial found a mean body weight reduction of 14.9% at 68 weeks — and crucially, the trial noted that weight loss was observed “from the first postrandomization assessment (week 4) onward,” with the rate of loss continuing to accelerate through approximately week 60. The STEP 1 data reinforces a fundamental pattern: the first four weeks are not where the weight comes off. The trajectory builds.
For standard Ozempic doses (0.5–1 mg), the SUSTAIN-1 trial in Diabetes Care found mean weight losses of 4.5% at 0.5 mg and 6.0% at 1 mg at 30 weeks, establishing the dose-response relationship and making clear that a significant portion of those losses accumulate well past the first month.
Why Patients With Type 2 Diabetes Lose Less
The Ghusn 2022 cohort documented a statistically significant difference: patients with type 2 diabetes lost 3.9% at 3 months versus 6.3% for those without diabetes (p=0.001). This gap persisted at 6 months: 7.2% versus 11.8% (p=0.005). Several mechanisms explain this: insulin resistance, different metabolic baselines, and, importantly, the fact that semaglutide’s glycemic effect in diabetics partially diverts the drug’s action toward blood sugar management. Non-diabetic patients using Ozempic off-label for weight loss are working with a biological advantage relative to diabetic patients in the weight loss context.
Factors That Shape Your Month 1 Results
No two patients respond identically to the 0.25 mg initiation dose. Several factors modulate what you experience in the first four weeks:
Starting weight. Heavier patients tend to lose more absolute pounds early, even if the percentage is similar. A 300-lb person losing 1.5% drops 4.5 lbs; a 180-lb person at 1.5% drops 2.7 lbs. The percentage metric is more clinically meaningful, but both are modest at this dose.
Diabetes status. Per the Ghusn 2022 data, having type 2 diabetes reduces early semaglutide-driven weight loss compared to metabolically healthy obese patients. This is a known dose-range effect, not a reason to abandon treatment.
Diet quality. Semaglutide suppresses appetite, but it cannot override dietary choices. Patients who use the reduced hunger signal to shift toward lower-calorie, higher-protein foods will accelerate early loss. Those who continue eating large, high-fat meals, which GLP-1 agonists slow the emptying of, potentially worsening nausea, may see both smaller weight losses and more side effects.
Side effect burden. GI symptoms are most common in the first four to eight weeks of each new dose level, per FDA Ozempic PI §6. In the pooled placebo-controlled SUSTAIN trials, nausea occurred in 15.8% of 0.5 mg patients and 20.3% of 1 mg patients versus 6.1% on placebo. These rates are from the therapeutic dose range; the 0.25 mg initiation dose typically produces lower rates. Patients with significant nausea or vomiting in month 1 may eat more cautiously than they otherwise would, which can paradoxically cause more early loss, or they may eat less nutritiously, which limits fat loss.
Exercise. Adding resistance training and moderate aerobic exercise does not dramatically change early weight loss percentages, but it preserves lean muscle mass during the caloric deficit, which matters for body composition and long-term metabolic rate. The STEP 3 trial in JAMA combined semaglutide with intensive behavioral counseling including exercise and found meaningfully better lean mass retention than medication alone.
GI Side Effects in Month 1: What’s Normal and When to Call Your Provider
Month 1 is the period of highest GI side effect burden for most Ozempic users. The FDA Ozempic prescribing information §6 states that “the majority of reports of nausea, vomiting, and/or diarrhea occurred during dose escalation”, meaning each step up in dose restarts a brief tolerance window. The first step (starting any dose at all) is week 1.
GI adverse event rates from pooled placebo-controlled SUSTAIN trials per FDA Ozempic PI §6:
| Adverse Effect | Placebo (n=262) | Ozempic 0.5 mg (n=260) | Ozempic 1 mg (n=261) |
|---|---|---|---|
| Nausea | 6.1% | 15.8% | 20.3% |
| Vomiting | 2.3% | 5.0% | 9.2% |
| Diarrhea | 1.9% | 8.5% | 8.8% |
| Abdominal pain | 4.6% | 7.3% | 5.7% |
| Constipation | 1.5% | 5.0% | 3.1% |
These rates are from the therapeutic dose range. At the 0.25 mg initiation dose, rates are lower, but symptoms are still possible. Overall GI adverse reactions occurred in 32.7% of 0.5 mg patients compared to 15.3% on placebo, per the same FDA source.
The good news: these symptoms are self-limiting. Per FDA PI §6, the GI effects peak early in treatment and taper as the body adapts, typically over four to eight weeks at any given dose level. Practical strategies that reduce GI burden: smaller, lower-fat meals; eating slowly; staying well-hydrated; avoiding carbonated beverages; and not lying down immediately after eating.
When to contact your provider: Persistent vomiting that prevents hydration, severe abdominal pain (which may signal pancreatitis, a rare but serious concern noted in FDA PI §5.1), or GI symptoms that do not improve after two weeks at the same dose warrant a call. Dose reduction or a longer initiation period is a legitimate clinical option, it does not mean treatment failure.
Maximizing Ozempic Weight Loss in Month 1
Month 1 is not where you drive dramatic results, it is where you build the foundation for results in months 2 through 6 and beyond. That said, several evidence-informed strategies optimize what you get from the initiation phase:
High-Protein Diet
Semaglutide reduces caloric intake but does not preferentially protect lean mass. Adequate protein, approximately 1.2–1.6 g per kilogram of body weight per day, counteracts muscle loss during the drug-driven caloric deficit. For a 200-lb (91 kg) patient, that is 110–145 grams of protein daily, achievable through lean meats, fish, eggs, Greek yogurt, and legumes. Protein also maximizes satiety per calorie, complementing semaglutide’s appetite-suppression mechanism.
Resistance Training
Two to three sessions per week of compound strength movements (squats, deadlifts, rows, presses) preserves metabolic muscle during fat loss. The AACE/ACE obesity clinical practice guidelines specifically recommend resistance training as a standard adjunct to GLP-1 pharmacotherapy.
Consistent Weekly Injection Timing
Semaglutide has a half-life of approximately seven days, meaning consistent weekly dosing builds steady-state plasma levels more effectively than irregular timing. Set a fixed injection day. Per FDA Ozempic PI §2, the day of the week can be changed as long as the minimum interval between doses is two days.
Managing Alcohol and Ultra-Processed Food
Alcohol contributes calories without nutritional value and may worsen GI symptoms in early titration. Ultra-processed foods, high in fat, refined carbohydrates, and sodium, are slower to empty from the stomach and tend to amplify nausea when gastric motility is already slowed by semaglutide.
What If You Don’t Lose Weight in Month 1?
Not losing measurable weight in the first four weeks on 0.25 mg is medically expected for many patients, not a sign of treatment failure. The 0.25 mg dose is explicitly a non-therapeutic dose per FDA PI §2.2. Before concluding the drug isn’t working, verify:
- Dose adherence: Are you injecting the full prescribed dose each week? Missed or under-delivered doses (caused by not holding the button for the full six seconds) reduce drug delivery.
- Injection technique: Confirmed subcutaneous delivery — abdomen (≥2 inches from the navel), upper thigh, or outer upper arm — per Novo Nordisk Instructions for Use.
- Diet context: If caloric intake has increased to compensate for reduced appetite (common in patients who misinterpret hunger cues), early loss will stall.
- Scale timing: Weight naturally fluctuates 2–5 lbs day-to-day due to water, food, and hormonal cycles. Weigh at the same time each morning, after using the bathroom and before eating, for an accurate trend.
If you are on 0.5 mg or higher and have seen zero weight loss after 8–12 weeks, that is worth discussing with your prescribing provider. The Ghusn 2022 data found a range of 3.6% to −14.3% at 3 months — a small subset of patients do not respond as expected, and non-response at therapeutic doses is a recognized clinical scenario that warrants evaluation (adherence, thyroid function, medications that counteract weight loss, etc.).
Beyond Month 1: What to Expect at 3, 6, and 12 Months
Month 1 is the runway, not the flight. Here is where the Ghusn 2022 Mayo Clinic data and the STEP 1 NEJM trial converge on a compelling picture:
| Milestone | Typical % Body Weight Loss | For 200-lb Start |
|---|---|---|
| Month 1 (0.25 mg initiation) | ~1–2% (estimated) | ~2–4 lbs |
| Month 3 | ~5.9% (Ghusn 2022 real-world) | ~11.8 lbs |
| Month 6 | ~10.9% (Ghusn 2022 real-world) | ~21.8 lbs |
| 30 weeks on 0.5 mg | ~4.5% (SUSTAIN-1, T2D patients) | ~9 lbs |
| 30 weeks on 1 mg | ~6.0% (SUSTAIN-1, T2D patients) | ~12 lbs |
| 68 weeks on 2.4 mg (Wegovy) | ~14.9% (STEP 1, NEJM) | ~29.8 lbs |
Month 1 estimate based on linearization from Ghusn 2022 three-month anchor; all other figures from primary sources. SUSTAIN-1 data from type 2 diabetes population; non-diabetic patients typically achieve higher percentage losses per Ghusn 2022.
The key insight: the 5.9% average at three months does not mean equal loss each month. Most evidence points to steeper loss in months 2 and 3, when you are at 0.5 mg or higher, than in month 1. Month 1 contributes 20–35% of the three-month total for most patients.
The 6-month figure of 10.9% from the Ghusn 2022 cohort is particularly meaningful for goal-setting: it suggests that doubling the duration roughly doubles the percentage loss in the Ozempic dose range, and that patients who reach dose escalation and maintain the medication see continued, compounding loss well past the first few months.
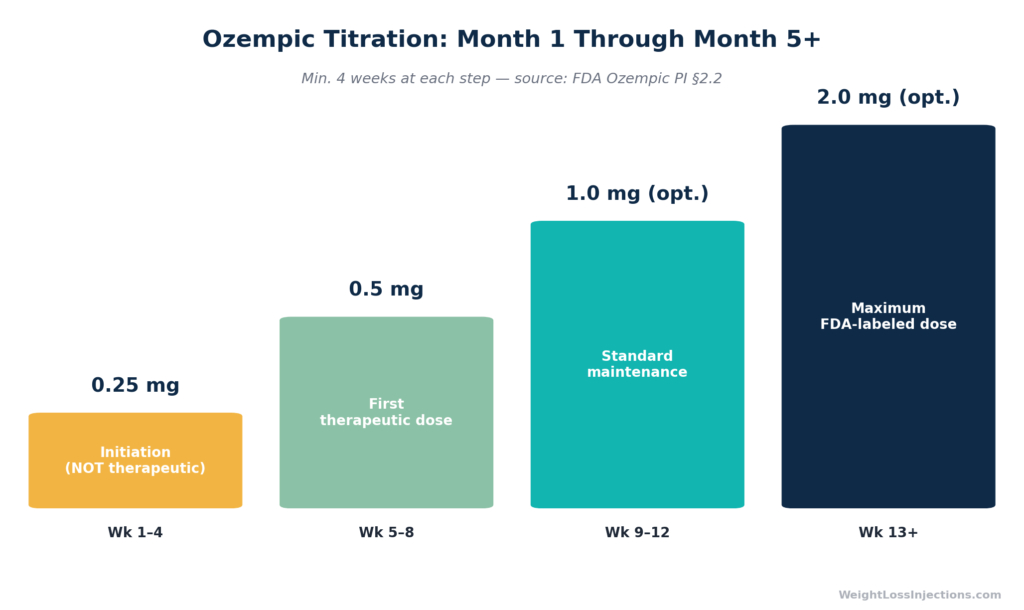
Ozempic Titration Timeline: Month 1 Through Month 5+
Getting Ozempic Through Telehealth at WeightLossInjections.com
Ozempic requires a licensed provider prescription. Telehealth has made that access substantially more efficient: at WeightLossInjections.com, patients complete a free online intake that covers medical history, current medications, BMI, and weight loss history. A licensed provider reviews your information and, if appropriate, issues a prescription along with a personalized titration plan tailored to your tolerance and goals.
The bundled telehealth program at WeightLossInjections.com is [$X/month], which includes [service detail], providing a streamlined option compared to managing medication access and provider care separately.
For context on the out-of-pocket medication landscape: Ozempic’s list price is $1,028/month, but patients without insurance can access it at $199/month for the first two fills through NovoCare Pharmacy (new patients, 0.25–0.5 mg, through June 30, 2026), then $349/month ongoing for 0.25–1 mg doses. A telehealth program that includes licensed provider oversight, dose adjustment guidance, and side effect management alongside medication access is particularly valuable during month 1’s tolerance phase, when most patients have questions about GI symptoms and titration timing.
Our take at WeightLossInjections.com: The most common reason patients abandon Ozempic in month 1 is unmanaged nausea combined with disappointment that the scale hasn’t moved dramatically. Both outcomes are predictable, and both are best addressed with a provider who sets accurate expectations before week 1 and is available when side effects hit in week 2. Month 1 exists to get you to month 3, which is where the clinical evidence shows meaningful results. The patients who succeed are the ones who understand that the first four weeks are the price of admission, not the payoff.
Frequently Asked Questions
How much weight can you lose in the first month on 0.25 mg Ozempic?
Most people lose approximately 1–5 lbs (roughly 1–2% of starting body weight) during the four weeks at the 0.25 mg initiation dose. This estimate is derived by back-extrapolating from the Ghusn 2022 Mayo Clinic cohort in JAMA Network Open, which recorded a mean loss of 5.9% at three months. Since month 1 is the sub-therapeutic initiation phase per FDA Ozempic PI §2.2 and most weight loss accelerates after escalation to 0.5 mg, a month-1 contribution of 1–2% is the realistic range. A 200-lb person at 1–2% loses 2–4 lbs; a 250-lb person loses 2.5–5 lbs. Some patients lose more, particularly if they make significant dietary changes in parallel.
Is losing 10 lbs in month 1 realistic on Ozempic?
For most patients, no. Losing 10 lbs in month 1 would require approximately 5% body weight reduction at the 0.25 mg initiation dose, but the Ghusn 2022 data shows that only 14.9% of patients even hit 10% at three months, not one. The 10 lb milestone is achievable, but typically happens around month 2–3, not month 1. A patient weighing 250+ lbs who makes substantial simultaneous dietary changes might approach 10 lbs by the end of month 1, but that would be at the upper end of the distribution. Setting this as a month-1 expectation is likely to produce frustration; setting it as a month-2 or month-3 target is more consistent with the evidence.
Why do people with diabetes lose less weight on Ozempic?
Patients with type 2 diabetes lose significantly less weight on semaglutide than non-diabetic patients at the same dose. The Ghusn 2022 Mayo Clinic cohort found a statistically significant gap: 3.9% at three months in T2D patients versus 6.3% in non-diabetic patients (p=0.001), and 7.2% versus 11.8% at six months (p=0.005). Several mechanisms contribute: insulin resistance itself impairs lipolysis; some diabetes medications (sulfonylureas, insulin) promote weight gain or counteract GLP-1-driven losses; and the metabolic milieu of diabetes blunts GLP-1 receptor sensitivity at some pathways. Non-diabetic patients using Ozempic off-label for weight management have a physiological edge in this context.
What if I see no weight loss in my first month on Ozempic?
Zero weight loss in month 1 at 0.25 mg is not necessarily a treatment failure. The dose is sub-therapeutic by FDA definition (PI §2.2), so meaningful loss is not guaranteed. Check injection technique (confirmed subcutaneous delivery, 6-second button hold per Novo Nordisk IFU), timing consistency, and whether dietary habits have meaningfully changed. Natural weight fluctuation of 2–5 lbs from water and food can mask early fat loss on the scale. If you have been on the 0.5 mg therapeutic dose for 8+ weeks and still see no movement, discuss with your provider, non-responders at therapeutic doses are uncommon but real, and a clinical evaluation for metabolic or adherence factors is warranted.
How does Ozempic compare to Wegovy in month 1 for weight loss?
Both start at the same 0.25 mg initiation dos, the Wegovy FDA prescribing information uses an identical four-week 0.25 mg initiation step. Month-1 differences are minimal. The divergence emerges in months 3–17: Wegovy’s therapeutic dose range extends to 2.4 mg weekly, versus Ozempic’s maximum of 2 mg. The STEP 1 trial at 2.4 mg showed 14.9% total weight loss at 68 weeks, compared to roughly 6–10% for Ozempic at its standard dose range per SUSTAIN trial data and Ghusn 2022. In month 1, you are on identical doses. The divergence is a months-3-through-12 story.
When does Ozempic weight loss really start to show?
Most patients report visible and measurable weight loss beginning in weeks 5–8, after escalating to the 0.5 mg therapeutic dose. Appetite suppression becomes more pronounced, portion sizes naturally decrease further, and the caloric deficit begins driving consistent fat loss. The STEP 1 trial noted that weight loss was observed “from the first postrandomization assessment (week 4) onward,” with the rate continuing to build. The Ghusn 2022 3-month anchor of 5.9% suggests that months 2 and 3 collectively contribute a larger share of early weight loss than month 1. For practical milestone planning: expect month 1 to deliver 1–2%, and months 2–3 to add another 3–4% on average.
This article is for educational purposes only and does not constitute medical advice. Ozempic is FDA-approved for type 2 diabetes; off-label use for weight management requires individualized provider assessment. WeightLossInjections.com’s editorial team reviews all content quarterly; last medical review: April 2026. Weight loss results vary by individual; trial averages represent population means, not guaranteed outcomes for any individual patient. Consult a licensed provider before starting any GLP-1 medication.

