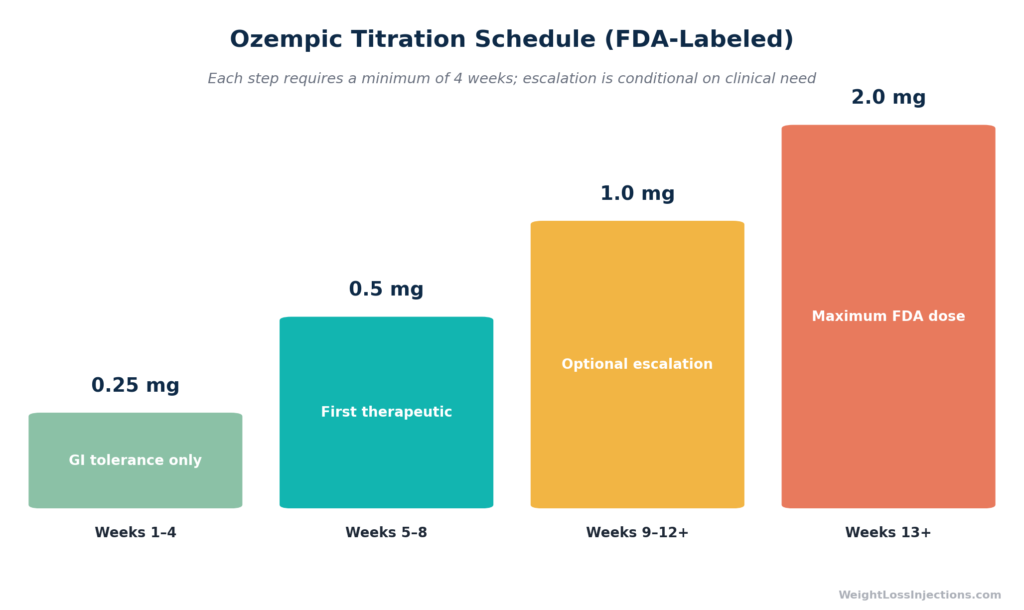
The FDA-labeled Ozempic starting dose is 0.25 mg once weekly for 4 weeks. This is not a therapeutic dose, it exists solely to build GI tolerance. Therapeutic dosing begins at 0.5 mg in week 5, with optional escalation to 1.0 mg and then 2.0 mg (the maximum). Minimum 4 weeks at each step. Never change your dose without your prescriber’s direction.
Safety notice: This article is for educational purposes only. Dosing decisions, including when to start, escalate, or hold, are your prescriber’s call. Never self-adjust your Ozempic dose. If you are experiencing severe side effects, contact your provider immediately.
Ozempic Titration Schedule
What Is the Ozempic Starting Dose?
The FDA-labeled starting dose of Ozempic (semaglutide) is 0.25 mg injected subcutaneously once weekly, maintained for the first four weeks of treatment. Per the FDA Ozempic Prescribing Information (2025), 0.25 mg is not intended for glycemic control, it is not a therapeutic dose. Its sole purpose is GI adaptation before reaching a dose that produces clinical effect.
This matters because skipping the initiation phase is one of the most common triggers for severe nausea, vomiting, and early discontinuation. Ozempic slows gastric emptying, and that effect is amplified abruptly at higher doses without the ramp-up. Four weeks at 0.25 mg is not a delay; it is your treatment starting correctly.
Ozempic is FDA-approved for glycemic control and cardiovascular/kidney risk reduction in adults with type 2 diabetes (FDA PI, 2025). It is not FDA-approved for weight loss, that indication belongs to Wegovy (semaglutide 2.4 mg). Off-label use for weight management is legal and common, but all dose decisions in that context are at clinician discretion. See our Ozempic for Weight Loss guide for more detail.
The Full Ozempic Titration Schedule
The FDA prescribing information specifies a four-step dosing ladder, with a minimum four-week duration at each step before any escalation (FDA Ozempic PI, 2025). Escalation beyond 0.5 mg is conditional, it applies only when additional clinical effect is needed, and is never automatic.
| Weeks | Dose | Purpose | Notes |
|---|---|---|---|
| 1–4 | 0.25 mg once weekly | GI tolerance only — not therapeutic | Do not escalate early |
| 5–8 | 0.5 mg once weekly | First maintenance/therapeutic dose | Hold here if GI effects are significant |
| 9+ (optional) | 1.0 mg once weekly | Escalation after 4+ weeks at 0.5 mg | If additional glycemic or clinical effect needed |
| 13+ (optional) | 2.0 mg once weekly | Maximum dose; escalation after 4+ weeks at 1.0 mg | Maximum recommended dose per FDA label |
This schedule is confirmed by the Mayo Clinic semaglutide dosage page and Drugs.com dosage guide, both of which align with the FDA PI’s stepwise titration. The governing principle is the same across all three: the slower the escalation, the lower the rate of GI-driven discontinuation.
Weeks 1–4: 0.25 mg
At 0.25 mg, most patients experience mild nausea, particularly in weeks one and two. This dose does not produce glycemic control, it is a physiological warm-up (FDA PI, 2025). Do not expect meaningful weight loss or significant appetite suppression.
What to expect: Mild queasiness within 24–48 hours of injection; occasional early satiety. Weight loss at this stage is not reproducible and is not a drug effect.
Weeks 5–8: 0.5 mg (First Therapeutic Dose)
The 0.5 mg dose is where appetite suppression becomes clinically noticeable. GI side effects tend to peak at the week-5 transition and diminish over subsequent weeks as tolerance builds. In the SUSTAIN trials, patients on semaglutide 0.5 mg achieved approximately 4–7% body weight reduction over 30 weeks, with 30–40% achieving ≥5% weight loss (Frontiers in Endocrinology, 2024). For some patients, 0.5 mg becomes the long-term maintenance dose, there is no requirement to escalate unless additional clinical benefit is needed.
What to expect: Noticeably earlier satiety, reduced food cravings, and for many patients a quieting of “food noise.” Nausea at the transition week typically softens within 10–14 days.
Optional Escalation: 1.0 mg
After a minimum of four weeks at 0.5 mg, your prescriber may increase to 1.0 mg weekly if additional glycemic control or clinical effect is warranted. In the SUSTAIN trials, the 1.0 mg dose produced approximately 7% body weight reduction over 30 weeks, with 45–60% of patients achieving ≥5% weight loss (Frontiers in Endocrinology, 2024). GI adverse reactions occurred in 36.4% of patients at 1.0 mg vs. 32.7% at 0.5 mg vs. 15.3% on placebo (FDA PI, 2025).
CKD patients: In type 2 diabetes with chronic kidney disease, the FDA label specifies a maintenance ceiling of 1.0 mg once weekly, escalation to 2.0 mg is not indicated in this population (FDA PI, 2025).
Optional Escalation: 2.0 mg (Maximum Dose)
The 2.0 mg weekly dose is the maximum FDA-labeled dose for Ozempic. Escalation from 1.0 mg requires at least four weeks at 1.0 mg and is not mandatory. In the SUSTAIN FORTE trial, patients escalating to 2.0 mg showed an incremental ~0.93 kg additional weight reduction and 0.23% additional HbA1c reduction vs. 1.0 mg (SUSTAIN FORTE, MDEdge / ADA 2021). The benefit is real but incremental; GI side effects increase modestly (34% at 2.0 mg vs. 30.8% at 1.0 mg) (FDA PI, 2025).
Our take at WeightLossInjections.com: For patients using Ozempic off-label for weight management, the titration schedule above is the starting framework, but your prescriber may modify timing at any step based on your GI tolerance and response. The off-label weight-loss context does not change the physiology: the GI system still needs the same ramp-up. Rushing titration to maximize weight loss faster almost always backfires.

Ozempic Pen Color/Dose Reference
Ozempic Pens: Which Pen for Which Dose
Not all Ozempic pens deliver all doses. Per Novo Nordisk’s prescribing resource and the GoodRx Ozempic dosage guide:
- Red-labeled pen: Delivers 0.25 mg and 0.5 mg only. Six total doses (four × 0.25 mg + two × 0.5 mg). This is the starter pen most new patients receive.
- Blue-labeled pen: Delivers the 1.0 mg dose. Four doses per pen.
- Yellow-labeled pen: Delivers the 2.0 mg dose. Four doses per pen.
Each pen is single-patient use and must never be shared (FDA PI, 2025). Store pens refrigerated before first use; after opening, pens may be kept at room temperature (up to 86°F) for up to 56 days.
Injection Technique — Brief
Inject subcutaneously once weekly into the abdomen (at least 2 inches from the navel), thigh, or upper arm (FDA PI, 2025). Rotate injection sites within the same region each week. Any day of the week at any time of day is acceptable — consistency in the chosen day matters more than which day. See our Ozempic Injection Sites guide for full technique detail.
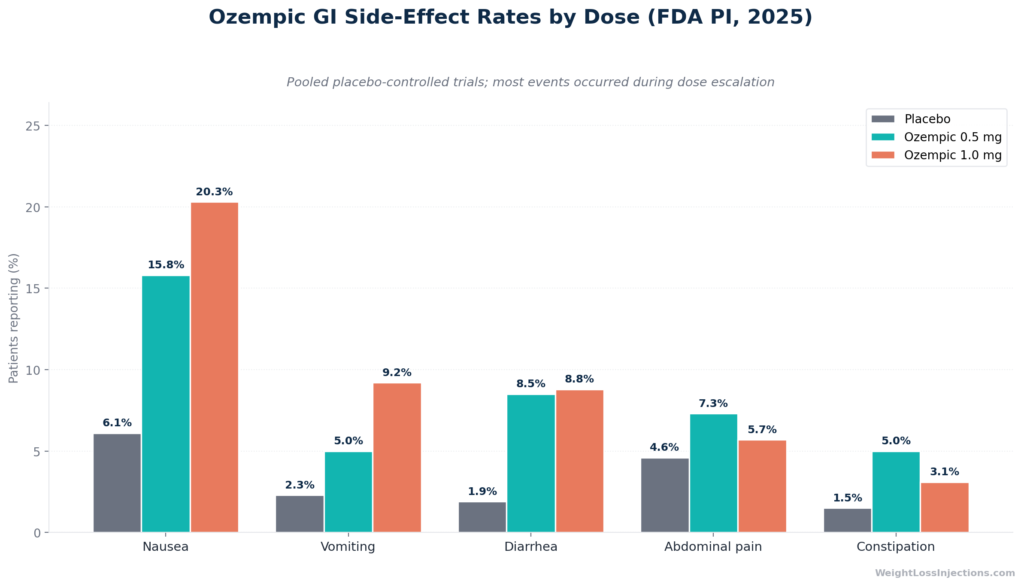
Managing GI Side Effects During Titration
GI side effects are the primary driver of titration pauses and early discontinuation. The FDA PI’s placebo-controlled trial data shows true incidence (FDA PI, 2025):
GI Side Effect Frequency by Dose
| Side Effect | Placebo | Ozempic 0.5 mg | Ozempic 1.0 mg |
|---|---|---|---|
| Nausea | 6.1% | 15.8% | 20.3% |
| Vomiting | 2.3% | 5.0% | 9.2% |
| Diarrhea | 1.9% | 8.5% | 8.8% |
| Abdominal pain | 4.6% | 7.3% | 5.7% |
| Constipation | 1.5% | 5.0% | 3.1% |
Source: FDA Ozempic Prescribing Information, 2025 — pool of placebo-controlled trials.
The majority of nausea, vomiting, and diarrhea occurred during dose escalation, not as a steady-state effect (FDA PI, 2025). Practical management: eat smaller, lower-fat meals; stay well hydrated; avoid lying down immediately after eating; reduce alcohol and carbonated beverages during the first two weeks at any new dose.
When to Pause Escalation
The FDA label does not specify formal hold criteria, but Mayo Clinic guidance recommends contacting your provider if GI symptoms are severe, persistent beyond 3–4 days, or interfere with eating or drinking. Staying at your current dose for an additional 4–8 weeks is a legitimate clinical decision.
Red-Flag Side Effects: Tell Your Provider Immediately
Certain symptoms go beyond expected GI discomfort and require urgent medical attention:
- Severe, constant abdominal pain — especially if it radiates to your back (potential pancreatitis signal)
- Persistent vomiting or diarrhea leading to signs of dehydration (dizziness, dark urine, inability to keep fluids down)
- Yellowing of skin or eyes, fever, right-upper-quadrant pain — potential gallbladder involvement
- Severe constipation with abdominal distension — potential ileus (intestinal blockage, added to the Ozempic label post-marketing)
These represent situations where continuing or escalating Ozempic without medical evaluation is not appropriate (FDA PI, 2025; Mayo Clinic, 2026).
Missed Ozempic Dose: Exact Rules from the FDA Label
The FDA Ozempic Prescribing Information (2025) states the missed-dose protocol clearly:
- Missed dose within 5 days: Take it as soon as you remember. Then resume your regular weekly schedule.
- Missed dose more than 5 days ago: Skip it entirely. Do not double up. Resume on your next scheduled day.
- Changing your injection day: You may switch the day of the week you inject, as long as at least 48 hours (2 days) have passed since your last dose.
This rule is confirmed identically by Drugs.com and Mayo Clinic. The 5-day window reflects semaglutide’s approximately one-week half-life, taking a missed dose late within that window still provides coverage; beyond it, blood levels from your previous dose have likely cleared.
T2D vs. Off-Label Weight-Loss Titration: What’s Different
The FDA titration schedule was established for type 2 diabetes management. When Ozempic is prescribed off-label for weight loss, the same stepwise schedule serves as the foundation, GI physiology is identical, but clinicians may apply it differently. Some hold patients at each step longer than four weeks if GI effects persist or if weight loss is already adequate at the current dose. Others follow the label schedule precisely. Weight-loss patients without diabetes may tolerate slower escalation more easily, as there is no A1c timeline driving urgency.
None of this reflects official FDA guidance, the label has no weight-management indication for Ozempic. These are real-world prescribing patterns. Your prescriber’s individualized plan governs your care.
WeightLossInjections.com editorial note: If you’re seeking Ozempic for weight loss through a telehealth provider, confirm your titration plan in writing at intake. Know which dose you’re starting at, how long you’ll stay there, what the escalation trigger is, and how to reach your provider if GI symptoms require holding. That clarity upfront prevents a lot of confusion later. Interested? Our GLP-1 intake quiz takes under 3 minutes, a licensed provider reviews your history within 24 hours and quotes you an all-in monthly cost of [$X/month] including [service detail].
When to Escalate vs. When to Hold
Escalate when you have tolerated your current dose for at least 4 weeks and have not yet achieved your target clinical response. Hold or delay if GI side effects remain significant after 2+ weeks, if weight loss is adequate at the current dose, or if you have CKD (1.0 mg ceiling). The FDA PI (2025) does not mandate escalation to 2.0 mg, it only requires that escalation not occur before four weeks at the prior dose. Many patients maintain long-term on 0.5 mg or 1.0 mg with no clinical reason to go higher.
Frequently Asked Questions
What is the starting dose of Ozempic?
0.25 mg once weekly for four weeks, per the FDA label. This is not a therapeutic dose, it does not produce meaningful glycemic control or weight loss. It exists solely for GI adaptation before the first maintenance dose (0.5 mg) begins at week five (FDA Ozempic PI, 2025).
How long do you stay on 0.25 mg Ozempic?
Four weeks, exactly. The label does not permit shortening this step. After week four, the dose increases to 0.5 mg unless your prescriber specifies otherwise (FDA PI, 2025; Mayo Clinic, 2026).
What if I miss my Ozempic starting dose?
Within 5 days: take it as soon as you remember, then resume your normal weekly day. More than 5 days past: skip it and resume on schedule, do not double up (FDA PI, 2025). If you’ve missed multiple weeks, contact your prescriber before resuming.
Can I start Ozempic for weight loss?
Ozempic is not FDA-approved for weight loss; that indication belongs to Wegovy. Prescribers may use it off-label when clinically appropriate. In the STEP 2 trial, semaglutide 1.0 mg produced approximately 7% body weight reduction at 68 weeks (STEP 2 / Journal of Pharmacy Technology, 2022). If weight loss is your primary goal and you qualify for Wegovy (BMI ≥30 or ≥27 with a comorbidity), discuss that label-approved option with your provider. See our Ozempic vs. Wegovy comparison.
What’s the Ozempic dose for CKD patients?
In type 2 diabetes with chronic kidney disease, the FDA label caps maintenance at 1.0 mg once weekly. After at least four weeks at 0.5 mg, the dose increases to 1.0 mg and does not escalate further (FDA PI, 2025). CKD patients face elevated dehydration risk if GI effects are severe, so close monitoring is especially important.
Key Sources
- FDA Ozempic Prescribing Information, 2025 — accessdata.fda.gov
- Mayo Clinic — Semaglutide (Subcutaneous Route) Dosage — mayoclinic.org
- Drugs.com — Semaglutide Dosage Guide — drugs.com
- SUSTAIN trial data — Semaglutide 0.5 mg and 1.0 mg weight outcomes — Frontiers in Endocrinology, 2024
- SUSTAIN FORTE — Semaglutide 2.0 mg vs. 1.0 mg — MDEdge / ADA 2021
- STEP 2 trial and STEP program review — Journal of Pharmacy Technology, 2022 — pmc.ncbi.nlm.nih.gov
- Novo Nordisk Ozempic Pen Reference — novomedlink.com
- GoodRx Ozempic Dosage Guide — goodrx.com
This article is for educational purposes only and is not a substitute for medical advice. WeightLossInjections.com’s medical team reviews content quarterly; last medical review: April 2026. If you are considering Ozempic or any GLP-1 medication, consult with a licensed provider about your individual medical history.

