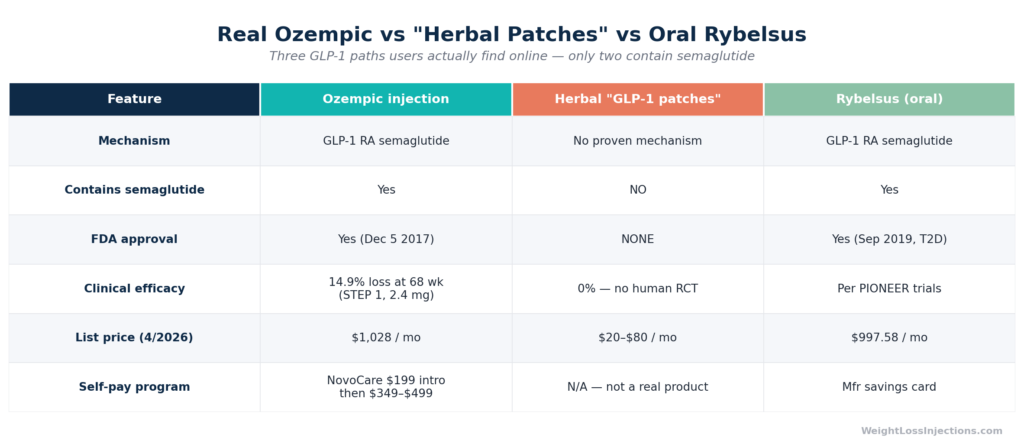
Real Ozempic Injection vs. Herbal ‘GLP-1 Patch’ vs. Oral Rybelsus, Head-to-Head Comparison
No FDA-approved Ozempic patch exists, and none is on the horizon for consumers. Viral ads for “GLP-1 patches” and “Ozempic patches” sell herbal supplements, berberine, garcinia, green tea, that contain no semaglutide and have no clinical evidence of weight loss. The reason a real semaglutide patch is biologically impossible with current consumer technology comes down to basic chemistry: semaglutide’s molecular weight of 4,113.58 Da is more than eight times the threshold at which molecules can cross human skin.
If you’re looking for medically supervised weight loss with GLP-1 therapy, the options that actually exist are injections and, for some patients, oral semaglutide, both available through licensed telehealth providers.
What Is the “Ozempic Patch”?
Search social media or budget wellness sites and you’ll find dozens of products with names like “GLP-1 Slim Patch,” “Ozempic-Style Weight Loss Patch,” or simply “Natural GLP-1 Patch.” The marketing is deliberate and misleading: these products borrow Ozempic’s cultural cachet, the drug’s name has become shorthand for rapid weight loss, while delivering none of its pharmaceutical substance.
A 2026 peer-reviewed analysis in Annals of Pharmacotherapy (White, Tai & Nuzi, 2026) examined 25 transdermal “natural GLP-1” patches and gels sold in the U.S. and found that none contained semaglutide, tirzepatide, or any FDA-approved GLP-1 receptor agonist. Instead, the average product contained seven herbal extracts, the most common being berberine (56% of products), pomegranate extract (44%), glutamine (44%), cinnamon (44%), and chromium (32%).
Real Ozempic is a prescription-only subcutaneous injection containing semaglutide, a synthetic analog of glucagon-like peptide-1 (GLP-1). It was FDA-approved on December 5, 2017 (NDA 209637) as an adjunct to diet and exercise for adults with type 2 diabetes, and later for cardiovascular risk reduction and, with its January 28, 2025 FLOW indication, chronic kidney disease. There is no patch version. There has never been a patch version. The distinction matters enormously because consumers spending money on fake patches are not just wasting money, they may be forgoing real, effective treatment during a window when that treatment could meaningfully improve their health.
Does an FDA-Approved Ozempic Patch Exist?
No. As of April 2026, there is no FDA-approved transdermal patch formulation of semaglutide or any other GLP-1 receptor agonist.
FDA Approval Status
The Ozempic prescribing information published by Novo Nordisk describes exactly three approved delivery formats: prefilled subcutaneous injection pens at 0.25 mg/0.5 mg, 1 mg, and 2 mg doses. No patch, cream, gel, or any transdermal formulation is listed. The FDA’s drug approval database (NDA 209637) reflects the same: all approved dosage forms are injection solutions.
Oral semaglutide exists, Rybelsus (approved September 2019 for T2D) and the Wegovy pill (launched January 2026 at $149–$299/mo NovoCare cash price), but these are swallowed tablets, not patches. The absorption mechanism is entirely different.
The FDA’s guidance on unapproved GLP-1 drugs used for weight loss is explicit: “Unapproved versions do not undergo FDA’s review for safety, effectiveness and quality before they are marketed.” Any product claiming to deliver GLP-1 effects through the ski, whether called a patch, serum, or gel, is making a claim the science cannot currently support.
Why No Patch? The Science of Skin Absorption
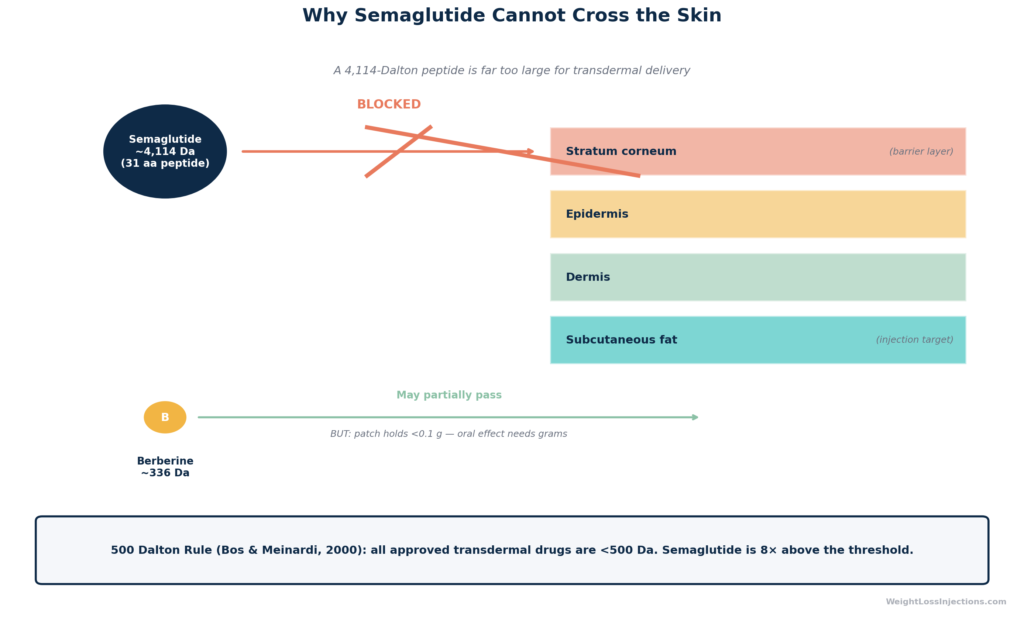
The barrier to a semaglutide patch is not regulatory or commercial, it is physical. Semaglutide is a 31-amino-acid peptide with a molecular weight of 4,113.58 g/mol per the Novo Nordisk prescribing information. Human skin is designed, among other things, to keep large molecules out.
The governing principle is the 500 Dalton rule, described by Bos & Meinardi (2000) in Experimental Dermatology (PubMed): virtually every molecule that successfully penetrates the stratum corneum and achieves systemic absorption through the skin has a molecular weight below 500 Da. The rule holds across contact allergens, topical dermatology drugs, and all approved transdermal delivery systems (nicotine patches, fentanyl patches, hormone patches). Semaglutide at 4,113.58 Da is more than eight times this ceiling.
To understand why, consider the stratum corneum, the outermost layer of skin, roughly 10–20 cell layers thick. It consists of dead, keratin-packed corneocytes embedded in a lipid matrix. Small, lipid-soluble molecules below ~500 Da can navigate between these cells or dissolve through the lipid matrix. Semaglutide is not only far too large; it is also highly hydrophilic (water-loving), because it is a peptide. As experts at UNSW and Macquarie University explain in a 2026 analysis, the skin is “highly lipophilic” by design, it absorbs oily, fat-soluble compounds and actively blocks water-loving ones. Semaglutide has both strikes against it: wrong size and wrong chemistry.
This is why Ozempic requires subcutaneous injection to bypass the skin entirely, delivering semaglutide directly into the tissue beneath, where it can be absorbed into the bloodstream. An injection achieves approximately 89% bioavailability. A topical patch would achieve essentially zero.
Skin Barrier Diagram: Why Semaglutide Cannot Cross the Stratum Corneum
Do Herbal “Ozempic Patches” Work for Weight Loss?
No. There is no human clinical trial evidence that any commercially sold herbal patch produces weight loss. This is not ambiguous, it is the conclusion of multiple independent reviews.
What’s Actually in These Patches
The White, Tai & Nuzi 2026 Annals of Pharmacotherapy analysis, the most comprehensive review of these products to date, found the following ingredients dominate the market:
| Ingredient | % of 25 Products | Oral dose studied | Oral result |
|---|---|---|---|
| Berberine | 56% | Up to 3 g/day for 1 year | Modest effect on weight and waist circumference |
| Green tea extract | 44% | Up to 2.4 g/day for 13 weeks | No effect on weight |
| Garcinia cambogia | 44% | More than 4 g/day for 17 weeks | No effect on weight |
| Cinnamon | 44% | Varies | Topical skin delivery not studied |
| Chromium | 32% | Varies | No transdermal weight loss evidence |
| Bitter orange (Citrus aurantium) | Present | Up to 54 mg synephrine for 8 weeks | No weight loss |
Two critical points from this table, explained by UNSW researchers Nial Wheate (Macquarie University) and Wai-Jo Jocelin Chan (UNSW Sydney):
- All evidence for these herbs comes from oral formulations — tablets, capsules, beverages. There is no clinical data supporting transdermal delivery of these ingredients for weight loss.
- Patches contain dramatically sub-therapeutic doses. Each patch holds less than 0.1 g of extract total. Berberine studies required up to 3 grams per day just to see a small oral effect. Even if the skin barrier did not exist, the dose is 30-fold too low to replicate those marginal results.
The Healthline review by pharmacist Kaylea Swearingen (updated April 2026) reaches the same conclusion: “There is no scientific evidence on the safety and effectiveness of ‘GLP-1 patches’… there is nothing to support that they work for weight loss.”
WeightLossInjections.com editorial note: We want to be direct about why this matters for you as a consumer. The search term “Ozempic patch” has reached millions of monthly searches precisely because real Ozempic can be difficult and expensive to obtain. Companies selling herbal patches are exploiting that gap. Spending $40–$80 on a patch is not a harmless experiment — it’s a delay in accessing a proven treatment path that, if you qualify, can produce meaningful, medically supervised weight loss.
Safety Risks: Not Just Ineffective, Potentially Harmful
Inefficacy alone would be concerning enough. But the White, Tai & Nuzi analysis documented serious safety signals across 25 products:
- 72% of products provided no health precautions or warnings on their labels
- Customer reviews across 9 products reported 32 skin-related adverse events (rashes, chemical burns, blisters, skin damage)
- 17 neurological complaints (headaches, tingling, anxiety, insomnia)
- 8 gastrointestinal adverse events (nausea, diarrhea)
- Contamination risk: Herbal products with no quality certification, zero of the 25 products carried USP or NSF certification, may contain heavy metals, undeclared prescription drugs, or adulterants. A separate Australian study cited by UNSW researchers found unregistered herbal products contaminated with warfarin, heavy metals, and other prescription drugs, substances that could be absorbed through broken or sensitive skin.
- Legal status: The Sage/Annals of Pharmacotherapy authors conclude these products are illegal under the Dietary Supplement Health and Education Act (DSHEA) of 1994, because DSHEA applies to products that are swallowed. Non-swallowed patches and gels fall outside that regulatory framework, creating a legal gray zone with essentially no oversight.
The FDA’s consumer alert on unapproved GLP-1 drugs broadly warns that unapproved products “could contain the wrong ingredients, contain too little, too much or no active ingredient at all or other harmful ingredients.” While this guidance primarily addresses compounded semaglutide, the principle applies directly to herbal products making GLP-1 efficacy claims: there is no regulatory backstop guaranteeing what is actually in the patch on your skin.
Real Ozempic: How It Works and What It Costs in 2026
How Ozempic Actually Produces Weight Loss
Semaglutide is a GLP-1 receptor agonist, it mimics the endogenous GLP-1 hormone released after eating. By activating GLP-1 receptors in the hypothalamus (the brain’s appetite regulation center), the pancreas, and the gut, semaglutide:
- Slows gastric emptying, reducing how quickly food leaves the stomach and prolonging satiety
- Reduces appetite and caloric intake via central nervous system signaling
- Improves glucose-dependent insulin secretion in patients with type 2 diabetes
- Reduces glucagon release, which helps lower blood glucose
The result is a consistent reduction in caloric intake that, sustained over weeks and months, produces clinically meaningful weight loss, not through any herbal stimulation, but through direct receptor pharmacology.
Proven Clinical Efficacy
In the STEP 1 trial published in the New England Journal of Medicine (2021), adults without diabetes treated with semaglutide 2.4 mg weekly (the Wegovy dose) achieved a mean 14.9% body weight loss at 68 weeks, compared to 2.4% with placebo. That is the benchmark figure driving public demand for GLP-1 therapy.
For Ozempic specifically (0.5 mg and 1.0 mg doses, studied in patients with type 2 diabetes), the SUSTAIN-1 trial published in Diabetes Care showed mean weight loss of 4.5% at 0.5 mg and 6.0% at 1 mg at 30 weeks. Real-world data from a Mayo Clinic JAMA Network Open analysis (Ghusn et al., 2022) found a mean 5.9% weight loss at 3 months in patients on semaglutide, across Ozempic doses.
No herbal patch has been tested in a rigorous randomized controlled trial for weight loss. The comparison is not between two products with different efficacy profiles; it is between a product with demonstrated clinical pharmacology and one with none.
2026 Pricing: What Ozempic Actually Costs
Ozempic’s list price is $1,028/month, up slightly from its prior $997/month price. Without insurance or assistance programs, this is prohibitive for most patients. However, several legitimate access pathways exist:
NovoCare self-pay pharmacy (Novo Nordisk’s direct program, verified April 2026):
- $199/month for the first two fills (0.25 mg and 0.5 mg pens) — introductory offer available through June 30, 2026, per GoodRx’s GLP-1 savings guide
- $349/month for ongoing 0.25 mg, 0.5 mg, and 1 mg doses
- $499/month for the 2 mg dose
Insurance coverage: Most commercial insurance covers Ozempic for on-label type 2 diabetes management with minimal cost-sharing. Coverage for off-label weight management is inconsistent and often requires prior authorization. The Medicare Part D 2026 out-of-pocket cap of $2,100 under the Inflation Reduction Act limits annual exposure for Medicare enrollees.
Rybelsus (oral semaglutide for T2D): List price $997.58/month, same across 3 mg, 7 mg, and 14 mg doses. Insurance coverage for T2D is available; without it, GoodRx lists Rybelsus 7 mg at approximately $1,229 without a coupon, though manufacturer savings cards can bring costs down for eligible patients.
2027 price reset: Novo Nordisk announced in February 2026 a planned list-price normalization to approximately $675/month for all semaglutide forms beginning in 2027, though this does not affect 2026 pricing.
Alternatives to Injections: What Actually Exists
Oral Semaglutide (Rybelsus)
For patients who cannot tolerate or decline injections, Rybelsus is the only FDA-approved oral semaglutide option currently in widespread use for type 2 diabetes. It requires specific administration conditions: taken on an empty stomach with no more than 4 oz of plain water, at least 30 minutes before eating. Oral bioavailability is approximately 0.4–1% due to peptide degradation in the gastrointestinal tract, which is why doses range up to 14 mg/day to achieve therapeutic serum concentrations. The Wegovy oral pill launched in January 2026 extends oral semaglutide options at higher doses for weight management, available through NovoCare at $149–$299/month depending on dose.
Neither pill is a patch. Both require swallowing, active GI absorption, and a valid prescription.
Emerging Research: Microneedle Patches (Not Available)
Legitimate research on needle-free semaglutide delivery does exist, but it is strictly preclinical.
A 2025 ADA abstract (1708-P, Tussey L. et al.) presented data on the MIMIX™ microarray patch (MAP), a device using microneedles (ultra-fine needles that penetrate only the superficial skin layers, not the subcutaneous tissue) to deliver semaglutide. The study was conducted in Göttingen minipigs, not humans. Key findings: comparable bioavailability to subcutaneous injection, with modestly faster early absorption and similar half-life (~84–87 hours). This is promising science.
Critically: This product is not available for human use. It has not entered human clinical trials. No regulatory approval exists or is pending for consumers. Any vendor claiming to sell a “microneedle Ozempic patch” to consumers today is not selling this research product, the MIMIX MAP is not commercially manufactured. They are selling something else entirely.
The distinction between preclinical research and a commercially available product is not a technicality. It is the difference between a concept tested in an animal model and a drug that has been proven safe and effective in thousands of human patients across multiple Phase 3 trials.
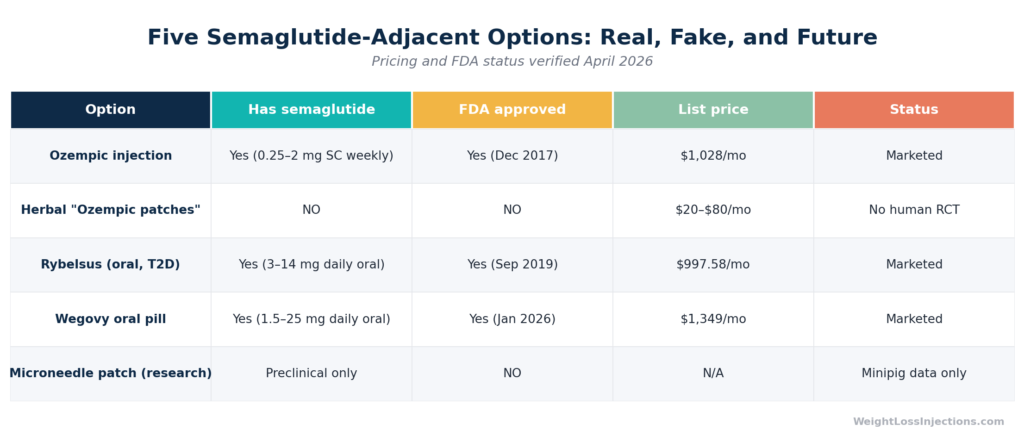
Ozempic Access Options: Pricing and Status Table
How to Get Real GLP-1 Therapy Like Ozempic
Obtaining a legitimate Ozempic prescription does not require an in-person appointment with a specialist. Telehealth has made the process significantly more accessible, while preserving the prescription requirements that exist for patient safety.
The Telehealth Process
A standard telehealth intake for Ozempic or GLP-1 therapy at a provider like WeightLossInjections.com involves the following steps:
- Online intake form — You provide your medical history, current medications, height, weight, diabetes status, and relevant comorbidities. Contraindications (history of medullary thyroid carcinoma, MEN2, prior pancreatitis, diabetic retinopathy, pregnancy) are screened at this stage.
- Provider review — A licensed physician or nurse practitioner in your state reviews your intake. This is a real clinical evaluation, not an automated rubber-stamp. The provider determines whether you meet criteria for Ozempic (T2D, cardiovascular risk reduction, CKD) or whether another GLP-1 such as Wegovy is more appropriate for your goals.
- Lab work if needed — Some providers request a recent A1C or metabolic panel, especially for patients with uncontrolled T2D or suspected contraindications.
- Prescription issued — If approved, a prescription is sent electronically to a licensed pharmacy of your choice or to the provider’s pharmacy partner.
- Medication shipped — [service detail]. Most patients receive their medication within 3–7 business days of prescription issuance.
WeightLossInjections.com offers GLP-1 telehealth services starting at [$X/month], [service detail]. Fill out the free intake form to see if Ozempic or another GLP-1 medication is clinically appropriate for you — provider review typically completes within 24 hours.
Clinical Eligibility Snapshot
Ozempic requires a prescription and a qualifying clinical picture. Most telehealth providers follow these general criteria (verify with your specific provider):
- On-label: Adults with type 2 diabetes requiring glycemic improvement, or adults with T2D and established cardiovascular disease or CKD
- Off-label weight management: BMI ≥ 30, or BMI ≥ 27 with at least one weight-related comorbidity (hypertension, dyslipidemia, sleep apnea, T2D, cardiovascular disease)
- Not eligible: Personal or family history of medullary thyroid carcinoma; Multiple Endocrine Neoplasia syndrome type 2; prior serious hypersensitivity to semaglutide; pregnancy or planning pregnancy within 2 months
The Real Risks of Fake Patches
We’ve covered the clinical picture. Here is the consumer protection picture.
Financial Harm
The average herbal patch costs $20–$80/month in retail channels. That amount won’t fund a month of Ozempic, but it represents money that could go toward NovoCare’s $199 introductory offer, a telehealth consultation, or lab work required for a legitimate evaluation. Over 6 months, $480 wasted on patches is $480 not applied to real care.
More dangerously: some fake “Ozempic patch” products retail at $150–$300/month, marketing themselves as comparable to the real drug. At those prices, patients could be funding a NovoCare prescription instead.
Physical Harm
The White, Tai & Nuzi analysis (2026) documented skin reactions including chemical burns and blisters from patch adhesives or ingredients. Contamination with undeclared prescription drugs (the UNSW researchers cite documented cases involving warfarin contamination in unregistered herbal products) is a real possibility. A patient on anticoagulants absorbing an undeclared dose of warfarin through a skin patch faces a serious bleeding risk.
Opportunity Cost and Diagnostic Delay
Patients self-treating with herbal patches delay the clinical evaluation that might identify type 2 diabetes, hypertension, or other conditions warranting real pharmacological intervention. Weight is not purely cosmetic — it is a metabolic marker. An unexamined BMI of 32 with borderline A1C is a medical situation, not a wellness optimization project.
How to Spot a Fake
Per the FDA’s guidance on unapproved GLP-1 products and the Healthline review (Swearingen, 2026), legitimate red flags include:
- Claims to “mimic Ozempic” or deliver “GLP-1 effects” via skin
- Available without a prescription
- No NDC number or FDA approval reference on packaging
- No USP or NSF quality certification
- Before/after weight loss photos without clinical trial citation
- Vague ingredient lists or “proprietary blends” with no dosing information
If a GLP-1 product does not require a prescription, it is not delivering semaglutide or any FDA-approved GLP-1 agonist. Period.
Conclusion
The “Ozempic patch” is a marketing fiction exploiting the most prominent weight-loss drug brand of the 2020s. No FDA-approved transdermal semaglutide formulation exists, and the biophysical barrier, a 4,113.58 Da molecular weight against a 500 Da skin-penetration ceiling, mean that passive transdermal delivery of semaglutide is not currently achievable without penetration-enhancing technology like microneedles, which remain strictly preclinical as of April 2026.
Herbal patches sold under GLP-1 branding contain no semaglutide, no FDA-approved active ingredients, no adequate dose of even their own listed herbal compounds, and carry meaningful safety and legal risks documented in peer-reviewed literature. The expert consensus, stated plainly by UNSW and Macquarie University researchers: “they do not work, and their safety is far from guaranteed.”
Our take at WeightLossInjections.com: The gap between what people want (a painless, easy, inexpensive path to weight loss) and what is clinically real (a prescription subcutaneous injection or pill requiring medical supervision) is exactly where predatory marketing lives. We focus on closing that gap with accurate information and legitimate access to GLP-1 therapy. If you qualify for Ozempic or a related GLP-1 medication, the NovoCare introductory pricing of $199/month through June 30, 2026 is the most affordable the drug has ever been for uninsured patients. That is the real opportunity — not a patch.
Frequently Asked Questions
Is there a real Ozempic patch available in 2026?
No. As of April 2026, no FDA-approved transdermal patch formulation of semaglutide (Ozempic’s active ingredient) exists. The only approved Ozempic delivery formats are subcutaneous injection pens, as documented in the Novo Nordisk prescribing information and FDA approval records (NDA 209637). Products marketed as “Ozempic patches” or “GLP-1 patches” do not contain semaglutide and are not FDA-approved. Preclinical microneedle research published at the ADA in 2025 shows early promise in animal models, but this technology is not available for human use.
Why can’t Ozempic be delivered through a skin patch?
The core barrier is molecular size. Semaglutide has a molecular weight of 4,113.58 g/mol per the Ozempic PI — more than eight times the ~500 Dalton threshold at which molecules can cross the skin’s stratum corneum. This 500 Dalton rule, established in pharmacology research and supported by every approved transdermal drug system (all are below 500 Da), means standard passive patch delivery cannot achieve meaningful semaglutide absorption. Additionally, semaglutide is a hydrophilic peptide, and skin preferentially absorbs lipophilic (fat-soluble) compounds. Subcutaneous injection bypasses these barriers entirely, which is why it is used.
Do herbal GLP-1 patches cause weight loss?
No. There is no human clinical trial evidence that any commercially available herbal patch produces weight loss. A 2026 peer-reviewed analysis of 25 such products (White et al., Annals of Pharmacotherapy) found that customer reviews reported no beneficial effects, and a Healthline review by a PharmD (Swearingen, 2026) confirms there is “nothing to support that they work for weight loss.” The herbal ingredients these patches contain, berberine, garcinia cambogia, green tea, have marginal evidence even from oral (swallowed) formulations at doses far exceeding what any patch can deliver, and no evidence from transdermal delivery at all.
How much does real Ozempic cost without insurance in 2026?
The list price for Ozempic is $1,028/month, per the most recent pricing as of April 2026. However, Novo Nordisk’s NovoCare self-pay program offers: $199/month for the first two fills (0.25 mg and 0.5 mg pens) through June 30, 2026; then $349/month for 0.25–1 mg doses, or $499/month for the 2 mg dose. These prices are available through eligible pharmacies with a valid prescription. Novo Nordisk has announced a 2027 list-price reset to approximately $675/month across semaglutide forms, though this does not affect current pricing.
How can I get prescribed Ozempic online?
Through a licensed telehealth provider, the process involves completing an online intake form covering your medical history, provider review of your eligibility (typically within 24 hours), and, if approved — a prescription sent to a licensed pharmacy that can ship to your home. WeightLossInjections.com offers GLP-1 telehealth services starting at [$X/month] with [service detail]. Eligibility broadly requires either a type 2 diabetes diagnosis, established cardiovascular disease, or a BMI ≥ 30 (or ≥ 27 with weight-related comorbidities) for off-label weight management. Contraindications include personal or family history of medullary thyroid carcinoma, Multiple Endocrine Neoplasia type 2, and pregnancy. Any legitimate telehealth platform will conduct a thorough clinical screening, if a site offers GLP-1 medications without a real prescribing evaluation, it is not operating legally.

