Ozempic Pancreatitis Anatomy & Symptoms Infographic
Pancreatitis is a rare but serious adverse event associated with Ozempic. The FDA Ozempic Prescribing Information (NDA 209637) reports 0.3 cases per 100 patient-years (PY) in the semaglutide arm versus 0.2 per 100 PY in comparators across SUSTAIN trials, a small numerical difference with no statistically significant increased risk confirmed in a 2024 meta-analysis of 34,000+ patients.
The hallmark symptoms, severe persistent abdominal pain radiating to the back, with or without vomiting, are distinct from routine Ozempic GI side effects. If these symptoms appear, stop Ozempic immediately and seek emergency care. Confirmed pancreatitis is a permanent contraindication to restarting semaglutide.
Safety notice: This article is for educational purposes only and does not constitute medical advice. Pancreatitis is a medical emergency. If you develop severe, unrelenting abdominal pain, with or without nausea and vomiting, seek emergency evaluation immediately and do not wait for a telehealth appointment.
What Is Pancreatitis?
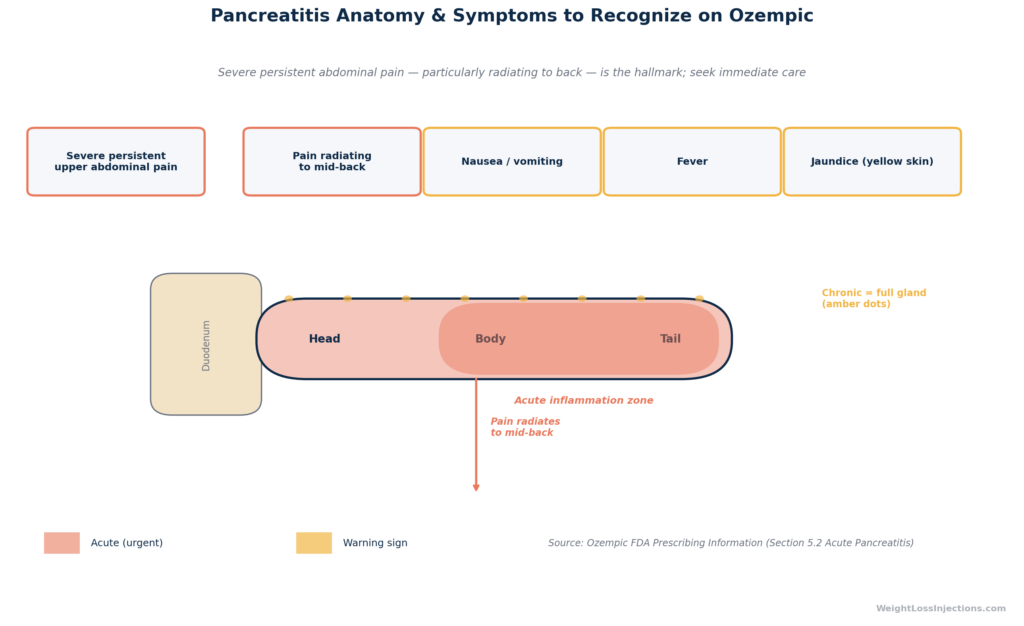
The pancreas is a roughly 6-inch glandular organ tucked behind the stomach in the retroperitoneal space, with its head nestled in the curve of the duodenum and its tail extending toward the spleen. It performs two essential jobs: its exocrine cells secrete digestive enzymes (lipase, amylase, proteases) through the pancreatic duct into the small intestine, and its endocrine islet cells secrete insulin and glucagon directly into the bloodstream to regulate blood glucose.
Pancreatitis is inflammation of the pancreas. When the organ becomes inflamed, those digestive enzymes can activate prematurely inside the gland itself, essentially beginning to digest the pancreatic tissue, causing pain, cellular damage, and systemic inflammatory response.
Acute pancreatitis is sudden-onset inflammation, typically resolving with treatment over days to weeks. Most cases are mild, but roughly 15–20% are severe, with organ failure, necrosis, and mortality rates in the 20–40% range for the severest forms, according to epidemiological reviews published in Gastroenterology.
Chronic pancreatitis is progressive, irreversible scarring of the pancreas caused by repeated episodes of inflammation or sustained injury, ultimately impairing both exocrine (digestive) and endocrine (insulin) function and predisposing to diabetes and pancreatic cancer.
Who Is Already at Higher Risk?
This matters enormously for Ozempic users, because the patient population most likely to be prescribed semaglutide, adults with type 2 diabetes (T2D) and obesity, already carries elevated baseline pancreatitis risk. Type 2 diabetes and obesity are each independent risk factors for pancreatitis. A 2019 meta-analysis in the British Journal of Surgery found that obesity was associated with a 74% higher risk of acute pancreatitis compared with normal weight. Hypertriglyceridemia, common in metabolic syndrome, is one of the top three causes of acute pancreatitis and can multiply personal risk by fourfold or more when triglycerides exceed 1,000 mg/dL. A prior episode of pancreatitis or a history of gallstones also markedly elevates recurrence risk. Understanding your personal baseline is the first step in an honest conversation with your prescriber.
Ozempic & Pancreatitis Risk: FDA Data
Clinical Trial Incidence
The most authoritative incidence data come directly from the FDA Ozempic Prescribing Information (NDA 209637, 2024 update). Across the SUSTAIN phase 3 program, seven randomized controlled trials enrolling thousands of adults with T2D, acute pancreatitis was reported at 0.3 cases per 100 patient-years in the semaglutide arm versus 0.2 cases per 100 patient-years in comparator arms. The FDA PI lists pancreatitis under “Warnings and Precautions” and states: “Acute pancreatitis, including fatal and non-fatal hemorrhagic or necrotizing pancreatitis, has been observed in patients treated with GLP-1 receptor agonists, including OZEMPIC.”
The table below summarizes pancreatitis incidence from the SUSTAIN trial program as reported in the Ozempic prescribing information and the SUSTAIN pooled safety analysis (Kristensen et al., Diabetes, Obesity & Metabolism, 2019):
| Trial / Pool | Semaglutide (cases/100 PY) | Comparator (cases/100 PY) |
|---|---|---|
| SUSTAIN 1–5 pooled | 0.3 | 0.2 |
| SUSTAIN 6 (CV outcomes) | 0.3 | 0.1 |
| Comparators include: placebo, sitagliptin, exenatide ER, insulin glargine, dulaglutide |
Source: FDA Ozempic PI, 2024; SUSTAIN pooled safety, Kristensen et al. 2019.
The numerical difference, 0.3 versus 0.2 per 100 PY, is small and not statistically significant in any individual trial. The FDA’s requirement to list pancreatitis as a warning reflects the class-level concern for GLP-1 receptor agonists established prior to Ozempic’s approval, as well as post-approval pharmacovigilance.
2024 Meta-Analysis Findings (PMID 38555109)
The most rigorous quantitative synthesis of GLP-1 RA and pancreatitis risk comes from a 2024 meta-analysis published in Diabetes Care (PMID 38555109), which pooled 34,000+ patients across randomized controlled trials evaluating semaglutide and other GLP-1 receptor agonists. Key findings:
- Odds ratio for pancreatitis with GLP-1 RA vs. comparator: 0.7 (95% CI: 0.5–1.1), indicating no statistically significant increased risk — in fact, a non-significant trend toward lower incidence.
- Results were consistent when stratified by dose (0.5 mg vs. 1.0 mg semaglutide) and by route of administration (subcutaneous vs. oral).
- The authors concluded that current RCT evidence does not support a causal link between GLP-1 receptor agonists and increased acute pancreatitis risk, but noted that post-marketing surveillance should continue given the biological plausibility of the GLP-1 receptor’s role in pancreatic exocrine function.
This finding is clinically important: it separates biologically plausible concern from demonstrated statistical risk. Pancreatitis is listed as a warning not because trials proved Ozempic causes it, but because (a) it has been reported in patients on the drug, (b) the drug’s mechanism includes pancreatic GLP-1 receptor activation, and (c) the consequences are serious enough to warrant vigilance.
Ozempic Pancreatitis Symptoms (Direct from Label)
Hallmark Signs
The FDA Ozempic Prescribing Information instructs prescribers to inform patients of the characteristic presentation. The PI advises that if pancreatitis is suspected, “OZEMPIC should be discontinued and appropriate management should be initiated.” The official label language on presentation describes:
“Persistent severe abdominal pain, sometimes radiating to the back and which may or may not be accompanied by vomiting.”
This phrase is the diagnostic anchor. Every word matters clinically:
- Persistent — pancreatitis pain does not resolve with antacids, position changes, or brief rest; it continues and often intensifies.
- Severe — not the mild cramping or transient discomfort of typical Ozempic GI side effects; patients describe it as one of the worst pains of their lives.
- Abdominal pain — typically epigastric (upper-center abdomen) or left upper quadrant.
- Radiating to the back — the retroperitoneal location of the pancreas means inflammation frequently manifests as back pain, often described as a boring or band-like sensation across the mid-back.
- With or without vomiting — nausea and vomiting are common accompaniments but are not required for the diagnosis.
Additional symptoms consistent with pancreatitis that appear in clinical gastroenterology guidelines include:
- Fever (low-grade initially; high fever suggests infected necrosis)
- Rapid heart rate (tachycardia from pain and systemic inflammatory response)
- Jaundice (yellowing of skin/eyes — suggests gallstone pancreatitis obstructing the bile duct)
- Abdominal distension and rigidity (severe cases)
Pancreatitis Symptoms vs. Common Ozempic GI Side Effects
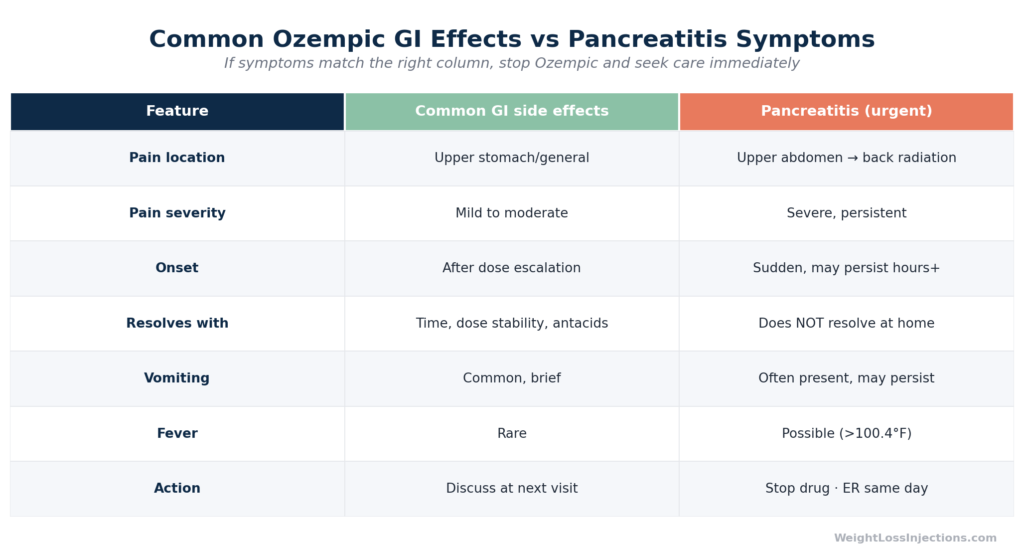
This distinction is the most practically useful element of this article. Routine Ozempic GI side effects — nausea, vomiting, diarrhea, constipation — are common, expected, and typically mild to moderate. Pancreatitis is rare and demands immediate emergency care. The table below maps the key distinguishing features.
Ozempic GI Side Effects vs. Pancreatitis Symptom Comparison Table
| Feature | Routine Ozempic GI Side Effects | Warning Zone | Pancreatitis — Emergency |
|---|---|---|---|
| Pain severity | Mild to moderate | Moderate | Severe (often 8–10/10) |
| Pain duration | Minutes to hours; transient | Persists >6 hours | Unrelenting; does not resolve |
| Pain location | Diffuse nausea, lower GI cramping | Epigastric / upper abdomen | Epigastric + radiates to back |
| Associated signs | Loose stools, mild vomiting | Persistent vomiting | Fever, rapid HR, jaundice (possible) |
| Timing | Peaks 24–48h post-injection (escalation) | Not necessarily injection-day | Not necessarily injection-day |
| Response to antacids | Partial relief common | Minimal relief | No meaningful relief |
| Action | Hydration, dietary adjustment | Same-day provider contact | Stop Ozempic — ER immediately |
Source: FDA Ozempic Prescribing Information, 2024; American College of Gastroenterology Pancreatitis Guidelines.
The single most important distinguishing feature is persistence and severity. Routine Ozempic nausea is unpleasant but typically settles within hours. Pancreatitis pain does not settle, does not respond to position changes or antacids, and is typically severe enough that patients cannot ignore it or push through their day.
Risk Factors & Who Is Most Vulnerable
Knowing your personal risk profile before starting Ozempic is a meaningful step, not because high risk means semaglutide is off the table, but because it should inform monitoring intensity and your threshold for seeking evaluation.
The FDA Ozempic prescribing information explicitly warns: “After initiating OZEMPIC, observe patients carefully for signs and symptoms of pancreatitis (including persistent severe abdominal pain, sometimes radiating to the back and which may or may not be accompanied by vomiting). If pancreatitis is suspected, OZEMPIC should be discontinued and appropriate management should be initiated; if pancreatitis is confirmed, OZEMPIC should not be restarted.”
Elevated-risk populations based on clinical evidence:
- Prior pancreatitis episode — The single strongest risk factor; recurrence rates after a first episode are 20–25% without addressing the underlying cause, per ACG guidelines. The PI does not list prior pancreatitis as an absolute contraindication, but the risk-benefit calculation deserves careful discussion with your prescriber.
- Gallstones or gallbladder disease — Gallstones are the most common cause of acute pancreatitis (40% of cases) (ACG guidelines). GLP-1 receptor agonists also slow gallbladder emptying, which can promote stone formation or sludge; the FDA PI for Ozempic lists cholelithiasis as a reported adverse event.
- Hypertriglyceridemia — Triglycerides above 500 mg/dL meaningfully increase risk; above 1,000 mg/dL the risk is substantial. A review in Pancreatology placed hypertriglyceridemia as the third leading cause of acute pancreatitis, responsible for 1–10% of cases depending on the population.
- Heavy alcohol use — The second most common cause of pancreatitis; alcohol is metabolized to toxic byproducts in pancreatic acinar cells and should be minimized on semaglutide.
- Obesity and T2D — Both independently increase baseline pancreatitis incidence, as noted above, meaning the drug-attributed numerator sits on a higher background denominator (British Journal of Surgery meta-analysis, 2019).
- Concurrent medications — Certain drugs (thiazide diuretics, some HIV antiretrovirals, azathioprine, valproic acid) have independent pancreatitis associations; drug interactions can compound risk.
What to Do If Symptoms Appear
Immediate Steps
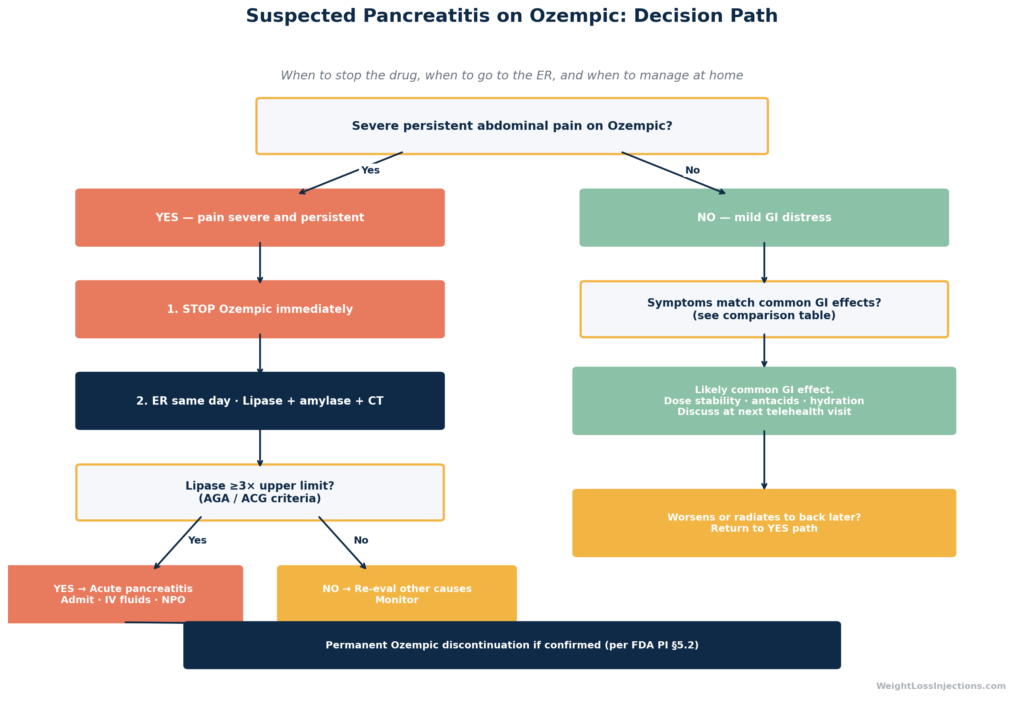
Do not wait and see. The hallmark FDA label language, persistent severe abdominal pain radiating to the back, is your action trigger.
- Stop Ozempic immediately. The FDA PI is explicit: if pancreatitis is suspected, discontinue semaglutide. Do not take your next scheduled dose.
- Go to the emergency room. Pancreatitis is confirmed by bloodwork and imaging — diagnosis cannot be made at home or by telehealth alone. Most hospitals use a serum lipase level ≥3× the upper limit of normal (ULN) as the primary diagnostic criterion, often combined with CT imaging when the diagnosis is uncertain or when complications are suspected (Atlanta Classification criteria for acute pancreatitis).
- Tell the ER team you are on Ozempic (semaglutide). This directly informs their clinical assessment and differential.
- Do not eat or drink while waiting for evaluation — most pancreatitis treatment protocols require early NPO (nothing by mouth) status to reduce pancreatic stimulation.
Diagnostic workup at the ER will typically include:
- Serum lipase (more specific than amylase; >3× ULN is diagnostic threshold per ACG guidelines)
- Serum amylase (less specific; may be normal in late-presenting cases)
- CT scan of the abdomen with contrast (confirms inflammation, identifies necrosis, rules out complications)
- Liver function tests and bilirubin (to evaluate for gallstone or biliary pancreatitis)
- Triglyceride level (to identify hypertriglyceridemia as a causative or contributing factor)
Hospital Treatment
Acute pancreatitis management is primarily supportive. The cornerstone of treatment is aggressive early IV fluid resuscitation, studies support lactated Ringer’s solution over normal saline for reduction of systemic inflammatory response, per ACG guidelines. Standard inpatient management also includes:
- Pain management — opioid analgesia is standard for severe pancreatitis; the goal is adequate pain control, not stoic endurance.
- NPO (nothing by mouth) — early, until pain and enzyme levels begin to normalize. Contrary to older practice, current guidelines support early oral feeding (within 24–48 hours when tolerated) rather than prolonged fasting, as enteral nutrition reduces complications and ICU stay (ACG guidelines).
- Nasojejunal tube feeding if oral intake cannot be resumed within 5–7 days.
- Monitoring for complications — necrosis, pseudocyst, abscess, organ failure (renal, respiratory, cardiovascular).
- Cholecystectomy — if gallstones are identified as the cause, early cholecystectomy during the same hospitalization reduces recurrence risk (ACG guidelines).
Recovery timeline for mild-to-moderate acute pancreatitis: Most patients are discharged within 3–5 days after lipase levels trend down, pain resolves, and oral intake is tolerated. Severe pancreatitis with necrosis or organ failure requires ICU-level care and carries a significantly prolonged course.
Prevention in Ozempic Users
Managing pancreatitis risk on Ozempic is not about avoiding the drug — it is about targeted monitoring and lifestyle measures that reduce modifiable risk factors.
Telehealth Monitoring Protocols
WeightLossInjections.com recommends the following surveillance framework for Ozempic users, particularly those with elevated baseline risk (prior pancreatitis, gallstones, high triglycerides):
- Week 4 labs: Fasting lipid panel (triglycerides), liver function tests (LFTs), amylase/lipase at baseline or at 4 weeks if initial levels were not obtained.
- Week 12 labs: Repeat lipid panel and LFTs to detect any medication-driven triglyceride change; GLP-1 receptor agonists can modestly affect lipid profiles, and any new hypertriglyceridemia should be addressed before it reaches the pancreatitis risk threshold.
- Weekly symptom check-in (first 8 weeks): During telehealth visits, providers should specifically ask about upper abdominal or back pain, not just nausea and GI symptoms. Pancreatitis pain is qualitatively different from GI side effects, but patients often initially attribute it to GI medication effects and delay seeking care.
- Patient education at initiation: Patients starting Ozempic should be explicitly told the pancreatitis warning language before they fill their first prescription — not buried in an insert, but verbalized by a provider: “If you ever develop severe persistent stomach pain that radiates to your back and doesn’t go away, stop the medication and go to the ER immediately.”
To set up a monitoring plan tailored to your medical history, WeightLossInjections.com offers telehealth consultations at [$X/month] for [service detail].
Lifestyle Risk-Reduction
- Limit alcohol — even moderate alcohol use can be a co-precipitating factor in patients with other risk factors. If you have a history of pancreatitis, alcohol cessation is strongly recommended.
- Follow a low-fat, low-refined-carbohydrate diet — high dietary fat directly stimulates pancreatic enzyme secretion and is the principal dietary driver of hypertriglyceridemia. Omega-3 fatty acids (fish, flaxseed) can actively lower triglycerides.
- Treat hypertriglyceridemia aggressively — if your triglycerides are elevated at baseline, your prescriber should consider fibrate therapy (fenofibrate or gemfibrozil) or prescription fish oil (icosapent ethyl/Vascepa) before or alongside Ozempic initiation.
- Address gallstones early — symptomatic gallstone disease in a patient about to start a GLP-1 RA warrants gastroenterology or surgical consultation before starting, not after.
When Can You Restart Ozempic?
The FDA Ozempic Prescribing Information is unambiguous: “If pancreatitis is confirmed, OZEMPIC should not be restarted.”
This is a permanent discontinuation, not a temporary hold. The concern is recurrence — a second episode of pancreatitis carries higher risk of severe complications, chronic pancreatitis, and irreversible organ damage. No dose reduction or extended washout period changes this guidance.
If the diagnosis is uncertain — for example, abdominal pain that was evaluated but where lipase was equivocal and imaging was non-specific — the restart decision must be made jointly by the patient and an experienced internist, endocrinologist, or gastroenterologist weighing the diagnostic certainty against the glycemic benefit. This is not a decision to make with a telehealth provider based on symptom history alone; it requires documented laboratory evidence.
Alternative Medications After Confirmed Pancreatitis
When Ozempic must be permanently discontinued, the prescribing team will need to address both glycemic control and, if weight management was the goal, alternative approaches. The FDA Ozempic PI does not mandate a specific alternative, but general options include:
- Metformin — first-line T2D agent with no pancreatic risk signal; typically well tolerated in pancreatitis history.
- SGLT-2 inhibitors (empagliflozin, dapagliflozin) — cardiovascular and renal benefits; no GLP-1–mediated pancreatitis mechanism.
- Insulin therapy — if significant beta-cell function has been lost (including from chronic pancreatitis impairing endocrine function).
- Note: Other GLP-1 receptor agonists (liraglutide, dulaglutide, tirzepatide) carry the same class-level pancreatitis warning and should generally be avoided if semaglutide-associated pancreatitis is confirmed.
Ozempic Pancreatitis Clinical Decision Flowchart
Our take at WeightLossInjections.com: Pancreatitis is the Ozempic warning that patients most frequently misunderstand in two opposing directions, either dismissing the risk entirely (“it’s so rare it doesn’t apply to me”) or catastrophizing it (“I can’t take this drug because my stomach hurts sometimes”). The FDA data and the 2024 meta-analysis (PMID 38555109) tell a nuanced story: no statistically increased risk in clinical trials (OR 0.7), but a biologically plausible mechanism, a non-zero rate in the semaglutide arm (0.3/100 PY), and consequences serious enough that the FDA requires explicit label warnings and prescriber counseling. The practical takeaway is calibrated vigilance: know the PI symptom language verbatim before you start (“severe persistent abdominal pain radiating to the back”), address modifiable risk factors (triglycerides, gallstones, alcohol), and do not tolerate weeks of upper abdominal pain hoping it will pass. Patients who do well on semaglutide long-term are the ones whose providers treated symptom education as a genuine pre-start conversation — not a box to check.
Frequently Asked Questions
Does Ozempic cause pancreatitis?
Clinical trials do not show a statistically significant increased risk. The 2024 meta-analysis (PMID 38555109) pooling 34,000+ patients found an odds ratio of 0.7 for pancreatitis with GLP-1 receptor agonists versus comparators, a non-significant trend in the other direction. The FDA Ozempic Prescribing Information reports 0.3 cases per 100 patient-years on semaglutide versus 0.2 on comparators across SUSTAIN trials. Pancreatitis is listed as a warning because it has been observed in patients on the drug (as it has been in patients with T2D and obesity independent of medication), the mechanism involves pancreatic GLP-1 receptors, and the consequences are serious. The current evidence does not establish Ozempic as a direct cause.
How soon after starting Ozempic can pancreatitis symptoms appear?
There is no fixed timeline. Cases have been reported within the first 30 days as well as after months of use, per pharmacovigilance data reviewed in the SUSTAIN safety analysis. Unlike nausea, which predictably peaks in the first 2–6 weeks of dose escalation, pancreatitis is not temporally locked to injection timing. This is one reason the symptom pattern matters more than the calendar: severe persistent pain radiating to the back is the signal, regardless of when it occurs.
Can I take Ozempic if I have gallstones?
Having gallstones significantly increases your baseline pancreatitis risk, as gallstone disease is the leading cause of acute pancreatitis (~40% of cases), per ACG guidelines. Additionally, GLP-1 receptor agonists may slow gallbladder emptying and are associated with cholelithiasis, per the FDA Ozempic PI. Ozempic is not absolutely contraindicated with a history of gallstones, but the decision requires an explicit risk-benefit conversation with your prescriber. Symptomatic or high-burden gallstone disease should typically be addressed surgically before starting a GLP-1 RA.
What labs confirm pancreatitis?
The primary diagnostic criterion is serum lipase ≥3× the upper limit of normal, which is more sensitive and specific than amylase in current practice, per ACG acute pancreatitis guidelines. Amylase may be elevated but can also be normal in late-presenting cases. CT of the abdomen with contrast is the definitive imaging study to confirm inflammation, assess severity, and identify complications (necrosis, fluid collections). No lab test can confirm pancreatitis from home or in a telehealth visit, if symptoms are consistent with pancreatitis, emergency evaluation is required.
Is pancreatitis from Ozempic reversible?
Acute pancreatitis, if recognized and treated promptly, is reversible in the majority of mild-to-moderate cases. Most patients recover full pancreatic function after a single episode of mild acute pancreatitis, per ACG guidelines. Severe cases complicated by necrosis, hemorrhage, or multi-organ involvement carry higher morbidity and can cause lasting damage, including progression to chronic pancreatitis and secondary diabetes. The key variable is time to treatment: severe, persistent symptoms that are ignored or managed at home rather than evaluated in the ER carry the highest risk of serious outcomes.
This article is for educational purposes only and is not a substitute for professional medical advice, diagnosis, or treatment. WeightLossInjections.com’s medical team reviews content quarterly; last medical review: April 2026. Always consult your licensed healthcare provider before making any changes to your medication regimen.

