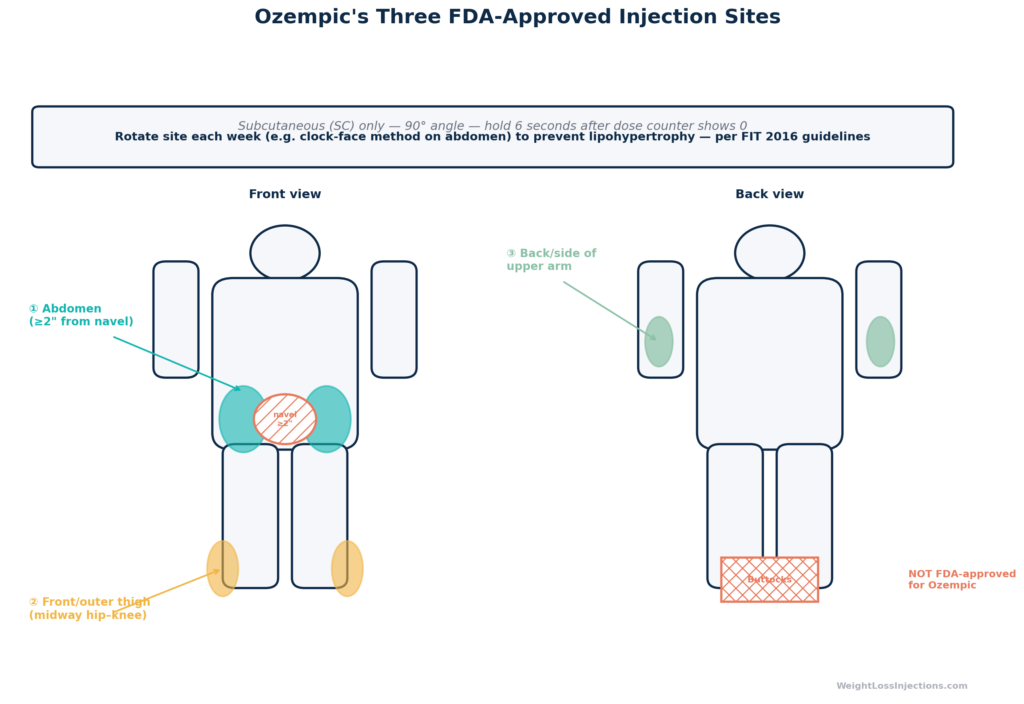
Ozempic Approved Injection Sites Body Diagram
Ozempic (semaglutide) is injected subcutaneously once weekly into one of three FDA-approved sites: the abdomen (at least 2 inches from the navel), the front or outer thigh (midway between hip and knee), or the back of the upper arm. All three sites produce equivalent drug absorption. Rotating within and among these zones every week prevents lipohypertrophy, the firm fatty deposits that form when the same spot is injected repeatedly. The buttocks are not an FDA-approved injection site for Ozempic.
Introduction: What You’re Actually Doing When You Inject Ozempic
Ozempic is a subcutaneous (under-the-skin) injection, not an intramuscular one. That distinction matters more than most patients realize. Hitting muscle instead of the subcutaneous fat layer can alter drug absorption, cause more pain, and lead to localized bleeding or bruising. Per FDA Ozempic prescribing information section 2.4 (Novo Nordisk, 2025), the injection must be administered subcutaneously in the abdomen, thigh, or upper arm. No other site is listed. No other route (IV, IM) is safe or approved.
Ozempic was FDA-approved on December 5, 2017 (NDA 209637) for type 2 diabetes management and cardiovascular risk reduction. It is also widely prescribed off-label for weight management. In both contexts, the injection sites, technique, rotation protocol, and dosing escalation schedule are identical. The drug does not know, or care, why you’re taking it. What it does care about is whether it’s delivered into the correct tissue layer at the correct depth.
The Ozempic Instructions for Use (Novo Nordisk, 2025) is the official patient-facing guide included in every Ozempic pen box. This article is built around that document, the FDA prescribing information, and published pharmacokinetic (PK) data on subcutaneous semaglutide absorption. Every factual claim below links to a primary source.
The Three FDA-Approved Ozempic Injection Sites
Per FDA Ozempic PI §2.4 (2025):
“OZEMPIC is administered subcutaneously in the abdomen, thigh, or upper arm. Rotate injection sites within the same region from one injection to the next.”
That sentence is the entire official statement on site selection. Here’s what it means in practice for each zone.
Abdomen (Stomach Area)
The abdomen is the most commonly used site and offers the largest surface area for rotation. The Ozempic Instructions for Use (Novo Nordisk, 2025) specifies the abdomen as a primary site, and Healthline’s semaglutide injection guide describes it as the most accessible location for most self-administering patients.
Key restriction: Stay at least 2 inches (5 cm) from the navel in all directions. The periumbilical area has a different subcutaneous fat depth and a denser concentration of nerve endings and blood vessels. Injecting too close to the navel increases pain, bruising, and the risk of inadvertently hitting tissue that does not absorb the drug efficiently. The Ozempic Instructions for Use diagram explicitly marks this exclusion zone.
Practical approach: Use a “clock face” rotation on the abdomen. Imagine the navel at the center. Divide the surrounding abdomen into eight positions (12 o’clock, 1:30, 3 o’clock, etc.) at least 2 inches out, then rotate through them sequentially. This allows 8+ distinct injection positions within a single region, important when the full rotation protocol (see below) keeps you in the abdomen for 4–8 weeks at a stretch before moving to another site.
Also avoid the waistband area, skin folds from tight clothing, any area near a belt buckle or waistband, and any section that has visible scarring, bruising, stretch marks, or tattoos. Per the Forum for Injection Technique (FIT) 2016 recommendations, injecting into damaged or altered skin tissue reduces absorption predictability.
Thigh (Front and Outer Surface)
The front and outer upper thigh, specifically the area midway between the hip and knee, is the second approved site. Per FDA Ozempic PI §2.4 and Ozempic Instructions for Use (Novo Nordisk, 2025), only the front and outer thigh surfaces are appropriate; the inner thigh has thinner subcutaneous fat and more pain-sensitive tissue.
Advantages: Like the abdomen, the thigh is easily self-administered without requiring a second person. Patients who have limited abdominal area due to scarring (e.g., from abdominal surgery) or who find abdominal injection uncomfortable often use the thigh as their primary site.
One caution: Injecting into the thigh immediately before vigorous lower-body exercise, running, cycling, heavy squatting, may theoretically alter local blood flow around the injection site. The Ozempic prescribing information section 12.3 (Novo Nordisk, 2025) does not specifically prohibit pre-exercise thigh injection, and the available pharmacokinetic evidence (see Absorption section below) shows no clinically meaningful site-to-site difference in drug exposure. But if you frequently exercise shortly after injecting, the abdomen may be a more predictable choice.
Do not inject into the inner thigh. Do not inject into the kneecap area. Stay in the midpoint zone between hip and knee.
Upper Arm (Back and Side)
The back and side of the upper arm is the third approved site. Per FDA Ozempic PI §2.4, this site is approved for self-injection, though most patients find it difficult to self-administer here without a mirror or assistant. A caregiver or household partner can administer this site reliably with the correct pinch technique.
The posterior-lateral upper arm has a defined subcutaneous fat layer that makes subcutaneous injection achievable, but the margin for error is smaller than in the abdomen. Injecting too far anteriorly (front of the arm) risks a shallower injection into thinner tissue; injecting too posteriorly and deep can approach the triceps muscle belly.
Important: This is the back of the upper arm (the “triceps” side), not the front (biceps). The front of the upper arm is not listed in the Ozempic Instructions for Use (Novo Nordisk, 2025).
What About the Buttocks?
The buttocks are not listed in the FDA Ozempic PI §2.4 (2025) as an approved injection site. This is a firm regulatory distinction, not a conservative suggestion. Do not inject Ozempic into the buttocks. The FDA approval applies specifically to the three sites listed above, and injection into unlisted sites is not supported by the pharmacokinetic data in the approved label.
Ozempic Dosing Schedule: The Official FDA Escalation Protocol
Ozempic’s dosing schedule is not discretionary. The FDA Ozempic PI §2.2 (Novo Nordisk, 2025) specifies a defined escalation pathway designed to allow gastrointestinal tolerance to develop before each dose increase.
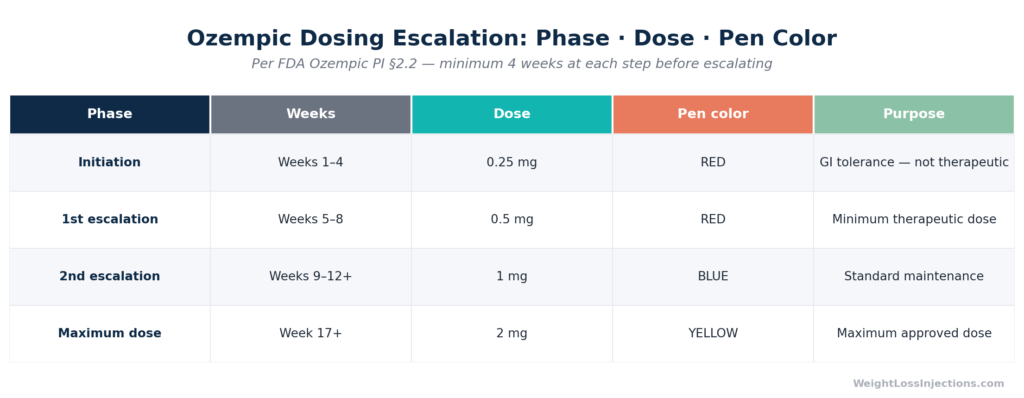
Ozempic Dosing Escalation Table: Pen Color, Dose, and Timeline
Initiation: Weeks 1–4 at 0.25 mg
Per FDA Ozempic PI §2.2 (Novo Nordisk, 2025):
“Initiate OZEMPIC with 0.25 mg once weekly for 4 weeks. The 0.25 mg dose is intended for treatment initiation and is not a therapeutic dose.”
The 0.25 mg initiation dose has one purpose: GI tolerance. It is not the dose that lowers HbA1c or produces meaningful weight loss. Patients who increase their dose before four weeks, trying to accelerate results, short-circuit this tolerance window and substantially increase their risk of nausea, vomiting, and diarrhea. From the FDA Ozempic PI §6.1 pooled placebo-controlled trial data, nausea occurred in 20.3% of patients at 1 mg versus 6.1% for placebo; premature escalation pushes patients toward the higher-dose GI burden before their system is ready.
The 0.25 mg pen is red-labeled and contains doses for both 0.25 mg and 0.5 mg in Novo Nordisk’s Ozempic pen lineup.
First Escalation: Weeks 5–8 at 0.5 mg
After four weeks at 0.25 mg, FDA PI §2.2 directs escalation to 0.5 mg once weekly. This is the minimum therapeutic dose for glycemic control. Patients using Ozempic off-label for weight management may notice appetite suppression beginning to strengthen at this dose.
Minimum time at this dose before any further escalation: 4 weeks.
Second Escalation: Weeks 9+ at 1 mg
If additional glycemic or weight-related control is needed after at least 4 weeks at 0.5 mg, FDA PI §2.2 permits escalation to 1 mg once weekly. This requires a new pen, the blue-labeled 1 mg Ozempic pen. For most patients using Ozempic for type 2 diabetes, 1 mg is the standard maintenance dose.
In the SUSTAIN-1 trial (Semaglutide vs placebo, NEJM 2017), semaglutide 0.5 mg produced 4.5% body weight loss and 1 mg produced 6.0% body weight loss at 30 weeks in a type 2 diabetes population, clinically meaningful differences that illustrate why the escalation step from 0.5 mg to 1 mg matters for real-world outcomes.
Maximum Dose: 2 mg
Per FDA PI §2.2, if the 1 mg dose does not provide sufficient glycemic control after ≥4 weeks, providers may escalate to 2 mg once weekly, the maximum approved dose. This requires the yellow-labeled 2 mg Ozempic pen.
The FDA Ozempic PI §6.1 notes that at 2 mg, GI adverse effects, particularly vomiting (noted in 9.2% at 1 mg, higher at 2 mg in some trials), become more prevalent. Escalation to 2 mg is at prescriber discretion based on clinical benefit versus tolerability.
Missed Doses
Per FDA Ozempic PI §2.1 (Novo Nordisk, 2025):
“If a dose is missed, administer OZEMPIC as soon as possible within 5 days after the missed dose. If more than 5 days have passed, skip the missed dose and administer the next dose on the regularly scheduled day.”
Never double-dose. Never inject two doses within a single week to compensate for a miss. The Ozempic PI §10 (Overdosage) notes that overdose management requires supportive care and that the approximately 1-week half-life of semaglutide means symptoms from excessive dosing can persist for days. The GoodRx pharmacist guide on missed GLP-1 doses aligns with this: a single missed dose, handled within the 5-day window, carries minimal risk.
Also per FDA PI §2.1: you may change your injection day as long as at least 48 hours have elapsed since your last dose. This permits permanent schedule adjustments without requiring a full 7-day gap.
Step-by-Step Injection Technique: 15 Steps from the Official Instructions for Use
The following 15-step protocol is drawn directly from the Ozempic Instructions for Use (Novo Nordisk, 2025). Do not improvise or shortcut these steps — each one exists to ensure full dose delivery, sterility, and subcutaneous (not intramuscular) placement.
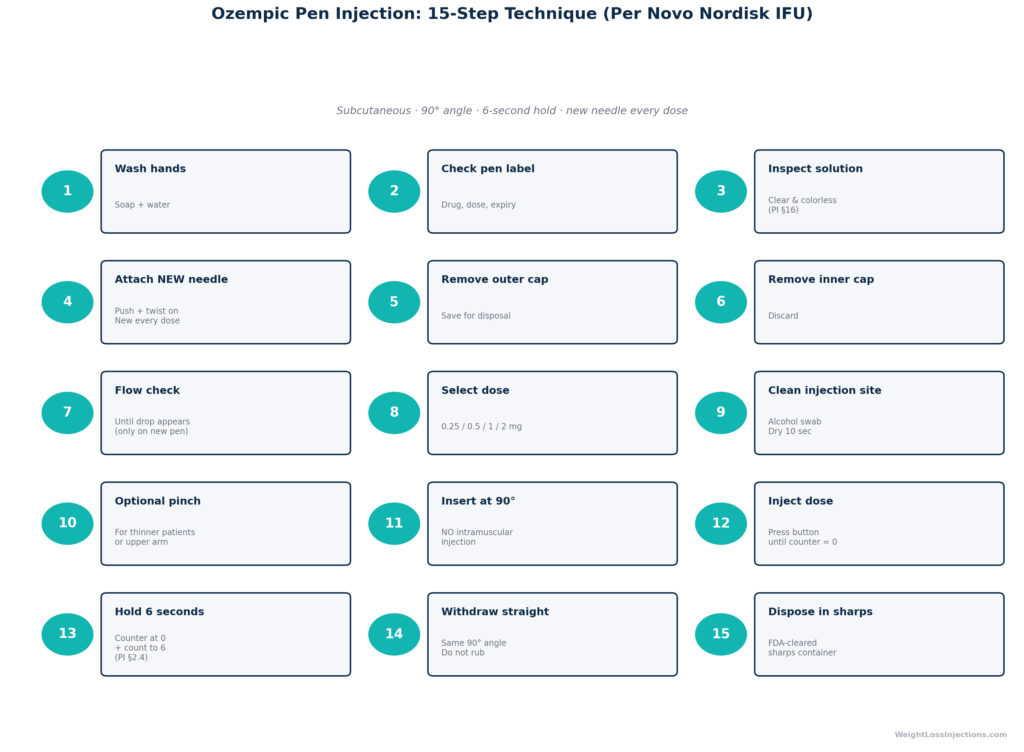
Ozempic Step-by-Step Injection Technique Infographic
Let’s highlight the steps that patients most commonly get wrong:
Step 7 — Flow check (priming the pen): Many patients skip this on the second or third use of a multi-dose pen, assuming it’s only for first use. The Ozempic Instructions for Use (Novo Nordisk, 2025) requires a flow check before every dose. Air in the needle tip can prevent full dose delivery.
Step 11 — 90° insertion: Subcutaneous injection into the abdomen and thigh is performed at a 90° (perpendicular) angle, per the Ozempic Instructions for Use (Novo Nordisk, 2025). Angling the needle reduces the effective depth into the subcutaneous fat layer and increases the risk of intramuscular injection in lean patients. Do NOT inject intramuscularly. Intramuscular injection of semaglutide is not approved and alters the absorption profile unpredictably.
Step 13 — The 6-second hold: This is the step patients most frequently shortcut. Per FDA Ozempic PI §2.4 (Novo Nordisk, 2025):
“After the dose counter shows 0, continue to hold down the dose button and count slowly to 6 before removing the needle from the skin.”
Releasing and withdrawing before 6 seconds allows the needle tip to evacuate the remaining dose from the injection channel back through the needle instead of into the tissue. Real-world dose shortchanging from premature needle withdrawal is a documented contributor to unexplained efficacy gaps in GLP-1 therapy. The 6-second hold is not optional.
Step 15 — New needle every dose: Per the Ozempic Instructions for Use (Novo Nordisk, 2025), a new needle must be used for every injection. Reused needles become dull, bent, and potentially clogged, which means incomplete dose delivery, increased injection pain, and higher infection risk. The Forum for Injection Technique (FIT) 2016 recommendations specifically flag needle reuse as a major contributor to lipohypertrophy and site complications across injectable therapies including GLP-1 agonists and insulin.
Rotating Ozempic Injection Sites: Why It Matters and How to Do It Correctly
Rotation is not a soft recommendation. It is a clinical requirement tied to a real, documented complication: lipohypertrophy.
What Is Lipohypertrophy?
Lipohypertrophy is the development of rubbery, thickened fatty deposits under the skin at repeatedly-injected sites. It occurs because subcutaneous fat cells hypertrophy (enlarge) in response to repeated local trauma from needle puncture and drug delivery. It is most extensively documented in insulin-dependent diabetic patients, but it applies to all subcutaneous injectables including semaglutide.
The FIT (Forum for Injection Technique) 2016 recommendations published in the Journal of Diabetes found lipohypertrophy in 38% of insulin-injecting patients studied across 18 countries — and showed that injecting into lipohypertrophic tissue was associated with significantly erratic drug absorption. Patients injecting into lipohypertrophic areas needed higher doses to achieve equivalent glucose control, and experienced more unexplained glycemic variability.
While the FIT data focused on insulin, the mechanism is site-agnostic: repeatedly injecting the same spot repeatedly creates tissue changes that reduce predictable subcutaneous absorption. For Ozempic specifically, the Novo Nordisk Ozempic Instructions for Use (2025) addresses this directly, mandating rotation within the same region from one injection to the next.
The FDA Ozempic PI §2.4 (2025) states: “Rotate injection sites within the same region from one injection to the next to reduce the risk of lipodystrophy.”
The Rotation Protocol
The Ozempic prescribing information §2.4 specifies rotation within the same region from one injection to the next — meaning you do not need to alternate between abdomen, thigh, and arm weekly. You can stay in one region (e.g., the abdomen) for multiple consecutive weeks, moving to a different spot within that region each time.
However, changing regions periodically gives each zone additional recovery time. A practical weekly rotation strategy:
| Week | Injection Region | Spot Within Region |
|---|---|---|
| 1 | Abdomen | 12 o’clock position |
| 2 | Abdomen | 3 o’clock position |
| 3 | Abdomen | 6 o’clock position |
| 4 | Abdomen | 9 o’clock position |
| 5 | Right thigh | Upper outer zone |
| 6 | Left thigh | Upper outer zone |
| 7 | Upper arm (right) | Posterior-lateral |
| 8 | Abdomen | 1:30 position (resume) |
Minimum spacing between adjacent injection points within a region: approximately 1 inch (2.5 cm), per FIT 2016 recommendations.
Always inspect the site before injecting. Per the Ozempic Instructions for Use (Novo Nordisk, 2025), do not inject into skin that is bruised, tender, red, scarred, or feels lumpy or hard. Those firmness cues are early indicators of lipohypertrophy. If you feel a thickened area, move at least an inch away and report it to your provider at your next visit.
Does Injection Site Affect Ozempic Absorption?
This is one of the most common clinical questions patients ask, and the answer may be more reassuring than you expect.
Novo Nordisk conducted pharmacokinetic studies of subcutaneous semaglutide to characterize whether the three approved sites produce meaningfully different drug exposure. Per data summarized in the Ozempic prescribing information §12.3 (Novo Nordisk, 2025), there is no clinically significant difference in semaglutide absorption across the abdomen, thigh, or upper arm. The area under the plasma concentration-time curve (AUC) and maximum concentration (Cmax) are comparable across all three sites.
This finding aligns with what Healthline’s semaglutide injection guide and GoodRx pharmacist guidance note for clinical practice: patients can choose their preferred site based on personal comfort, ease of self-administration, and skin condition — not based on expectations that one site will produce faster or stronger results.
The practical implication: choose the site that gives you the most consistent, comfortable, low-stress injection experience. Consistency in injection technique matters more than site selection for achieving stable semaglutide plasma levels.
Storage and Handling: What the FDA PI Actually Says
Incorrect storage degrades semaglutide before it ever reaches your injection site. Per FDA Ozempic PI §16 (Storage and Handling, Novo Nordisk, 2025):
Before first use (new, unused pen):
- Store in the refrigerator between 36°F and 46°F (2°C to 8°C).
- Do not freeze. Do not store near the freezer compartment or against the back wall of a refrigerator. Frozen semaglutide is degraded semaglutide.
- Keep the pen cap on when not in use. Protect from light.
After first use (pen in active use):
- May be stored at room temperature (up to 77°F / 25°C) or in the refrigerator for up to 56 days (8 weeks) from the date of first use.
- Per FDA PI §16, discard any remaining solution after 56 days even if doses remain.
- Keep away from heat sources and direct sunlight.
Before injection, inspect the solution: Per FDA PI §16 and the Ozempic Instructions for Use (Novo Nordisk, 2025), Ozempic should be clear and colorless to very slightly yellow. Do not use if the solution is cloudy, discolored, or contains visible particles. A cloudy or discolored pen is not just degraded — injecting it could cause a local reaction.
Needles: Store separately from the pen. Do not attach a needle to the pen between doses — this prevents air from entering the pen and contaminating the remaining solution. Per Ozempic Instructions for Use (Novo Nordisk, 2025), the needle should only be attached immediately before injection.
The Novo Nordisk Ozempic patient website summarizes these storage rules in patient-friendly language and confirms the 56-day post-first-use limit.
Common Mistakes and How to Fix Them
1. Injecting Too Early (Premature Needle Withdrawal)
Withdrawing the needle before the dose counter reaches 0 and before holding 6 full seconds is the most common technical error. The drug is not fully delivered. You have lost part of your dose. Per FDA PI §2.4: hold the dose button fully depressed and count to 6. If you are unsure whether you got the full dose, do not inject again — contact your provider.
2. Reusing Needles
Ozempic Instructions for Use (Novo Nordisk, 2025) is explicit: one needle per injection. Needle reuse blunts the tip (increasing trauma and pain), risks clogging (incomplete dose), and increases infection risk. The FIT 2016 recommendations also identify needle reuse as a primary driver of lipohypertrophy across injectable therapies.
3. Not Rotating Sites
Returning to the same injection spot week after week because “it’s easy” or “it doesn’t hurt there anymore” is exactly the problem, the absence of pain at a previously injected spot can indicate early lipohypertrophy, not that the spot is ideal. That numbed, hardened tissue absorbs drug less reliably per FIT 2016.
4. Wrong Angle: Angling Instead of Perpendicular
Subcutaneous injection for Ozempic is performed at 90°. Tilting the needle creates a shallower entry that may place the drug in the dermis (skin layer) rather than subcutaneous fat — causing a visible bleb, more pain, and reduced or erratic absorption. The Ozempic Instructions for Use (Novo Nordisk, 2025) diagrams 90° insertion for all sites.
Exception: Very lean patients with minimal subcutaneous fat may need to pinch skin before inserting at 90° to ensure the needle enters fat rather than muscle. Per the FIT 2016 recommendations, a skin pinch technique is appropriate for lean adults at thinner-fat sites like the thigh and upper arm to avoid intramuscular injection.
5. Injecting Into Compromised Skin
Injecting over bruises, scars, tattoos, stretch marks, varicose veins, or skin affected by psoriasis or eczema reduces absorption predictability and increases local irritation. The Ozempic Instructions for Use (Novo Nordisk, 2025) specifies avoiding these areas explicitly.
6. Skipping the Flow Check
The flow check confirms the needle is patent (not clogged) and the pen mechanism is working before you inject. Skipping it means discovering the problem mid-injection — or worse, not discovering it at all and believing you have received your dose. Per Ozempic Instructions for Use (Novo Nordisk, 2025), perform the flow check before every injection. If no drop appears after 6 attempts, do not use that pen — contact your pharmacy.
Injection Sites and Weight Loss: Is There a Difference?
For patients using Ozempic off-label for weight management, the majority of readers asking about injection sites, a common question is whether site selection affects weight loss outcomes specifically.
The direct answer, based on the Ozempic PI §12.3 pharmacokinetic data (Novo Nordisk, 2025), is no: the AUC and Cmax values are equivalent across the three approved sites. The drug reaches the same plasma concentrations regardless of whether you inject into your abdomen, thigh, or upper arm. Because the weight-loss mechanism of semaglutide operates through circulating GLP-1 receptor agonism, affecting appetite centers in the brain and gastric emptying rate, the site of subcutaneous absorption does not confer a directional weight-loss advantage.
What does affect weight loss outcomes at the injection level is dose delivery accuracy. Premature needle withdrawal, needle reuse leading to clogged delivery, and injection into lipohypertrophic tissue all reduce how much drug actually enters circulation per dose. In aggregate, these errors can produce the same real-world result as being on a lower dose than prescribed, reduced appetite suppression and slower weight loss.
The SUSTAIN-1 trial (NEJM 2017) documented 6.0% body weight loss at 30 weeks on semaglutide 1 mg, while the Ghusn 2022 real-world study in JAMA Network Open (Mayo Clinic) found 5.9% loss at 3 months on semaglutide in a clinical population. Real-world outcomes track trial outcomes reasonably closely when patients use correct injection technique consistently. Technique errors are one documented reason real-world outcomes diverge downward from trials.
Patients pursuing weight management with Ozempic through a telehealth provider should receive explicit injection training as part of their onboarding, not just a prescription. At WeightLossInjections.com, our telehealth program includes [service detail] to ensure patients are properly trained on technique, rotation, and dosing escalation before their first injection.
WeightLossInjections.com Editorial Note: The three-site rotation protocol sounds simple, and it is, once you build the habit. What we see in patients who plateau or report erratic appetite suppression is often a technique issue rather than a drug failure: missed flow checks, abbreviated holds, or repeated use of the same abdominal spot. Before concluding a dose isn’t working, verify your technique against the 15-step protocol above and the official Ozempic Instructions for Use (Novo Nordisk, 2025). If you’re managing your Ozempic prescription through our platform, your assigned clinician can review injection technique directly during a telehealth visit. Telehealth-managed Ozempic through WeightLossInjections.com starts at [$X/month], including prescriber consultations, lab review, and ongoing dose management.
Frequently Asked Questions
Where are the best Ozempic injection sites?
Per the FDA Ozempic prescribing information §2.4 (Novo Nordisk, 2025), the three approved sites — abdomen, front/outer thigh, and back of the upper arm — are clinically equivalent in terms of semaglutide absorption. The “best” site is the one you can consistently inject into correctly, at 90°, with proper rotation. Most patients prefer the abdomen for its large surface area and ease of self-administration. Patients with abdominal scarring often use the thigh. The upper arm is useful when a partner can assist. The buttocks are not an approved site per the FDA label.
Does injection site affect Ozempic absorption or weight loss?
No clinically significant difference in semaglutide absorption has been observed between the three approved sites, per pharmacokinetic data in the Ozempic PI §12.3 (Novo Nordisk, 2025). AUC and Cmax are comparable across abdomen, thigh, and upper arm. Site selection does not affect weight loss speed or magnitude. What does affect drug delivery accuracy is injection technique, specifically the 6-second hold per FDA PI §2.4, avoiding lipohypertrophic tissue per FIT 2016, and using a new needle every dose per Ozempic Instructions for Use (Novo Nordisk, 2025).
How do I rotate Ozempic injection sites?
Per FDA Ozempic PI §2.4 (Novo Nordisk, 2025), rotate to a different spot within the same region from one injection to the next. You do not need to change regions every week, you can stay in the abdomen for multiple weeks while moving between clock-face positions at least 1 inch apart. When you eventually move to the thigh or upper arm, use a new zone within that region. Avoid any area with lipohypertrophic lumps, bruising, or scarring. Track your rotation either in a written log or with a rotation tracking app. Consistent rotation prevents lipohypertrophy per FIT 2016 recommendations.
What if I miss an Ozempic dose?
Per FDA Ozempic PI §2.1 (Novo Nordisk, 2025): if your missed dose is within 5 days of the scheduled day, take it as soon as possible and resume your regular weekly schedule. If more than 5 days have passed, skip it and take the next dose on your scheduled day. Never double-dose to compensate, this is explicitly contraindicated in the Ozempic PI §10. If you’ve missed 2 or more weeks, contact your prescriber before resuming, you may need to restart at 0.25 mg or 0.5 mg to rebuild GI tolerance per GoodRx pharmacist guidance.
How should I store Ozempic pens before and after first use?
Per FDA Ozempic PI §16 (Novo Nordisk, 2025): unused pens must be refrigerated at 36–46°F (2–8°C); never frozen. Once you have used a pen for the first time, it may be stored in the refrigerator or at room temperature up to 77°F for up to 56 days from the date of first use. After 56 days, discard even if doses remain. Always inspect the solution before each injection, clear and colorless is correct; cloudy or discolored means do not use. Store pens without a needle attached, per Ozempic Instructions for Use (Novo Nordisk, 2025).

