Ozempic Plasma Decay Chart Over 5 Weeks Post–Last Dose
Ozempic (semaglutide) has an elimination half-life of approximately one week. That means it takes roughly five weeks, five half-lives, for the drug to fall below 3% of its peak steady-state level after your last injection. Therapeutic effects, side effects, and detectable plasma levels all taper over that same five-week window.
No physiologic factor, including kidney disease, liver disease, or advanced age, meaningfully speed or slow this process per the FDA label.
What Is Ozempic and How Does It Work?
Ozempic is the brand name for injectable semaglutide, a glucagon-like peptide-1 (GLP-1) receptor agonist approved by the FDA on December 5, 2017 (NDA 209637) for glycemic control in adults with type 2 diabetes. In January 2025, the FDA added a chronic kidney disease indication based on the FLOW trial results. The drug has also become widely prescribed off-label for weight loss, though its sister medication Wegovy (semaglutide 2.4 mg) carries the formal weight-management approval.
Semaglutide works by mimicking GLP-1, a hormone released after eating. It stimulates insulin secretion, suppresses glucagon, slows gastric emptying, and reduces appetite via central nervous system receptors. Per the FDA Ozempic prescribing information (2025), semaglutide is structurally modified from native GLP-1, with amino acid substitutions at positions 8, 26, and 34 plus a C18 fatty acid side chain, to dramatically extend its half-life. Native GLP-1 clears the body in minutes; semaglutide lingers for weeks.
That prolonged exposure is the pharmacologic rationale for once-weekly dosing. It is also why understanding the elimination timeline matters: when you stop Ozempic, its effects do not vanish overnight. They taper gradually, which has real implications for blood sugar management, weight rebound, pregnancy planning, and safely switching to another GLP-1 medication.
Ozempic’s Half-Life and Elimination Timeline
The ~1-Week Half-Life Explained
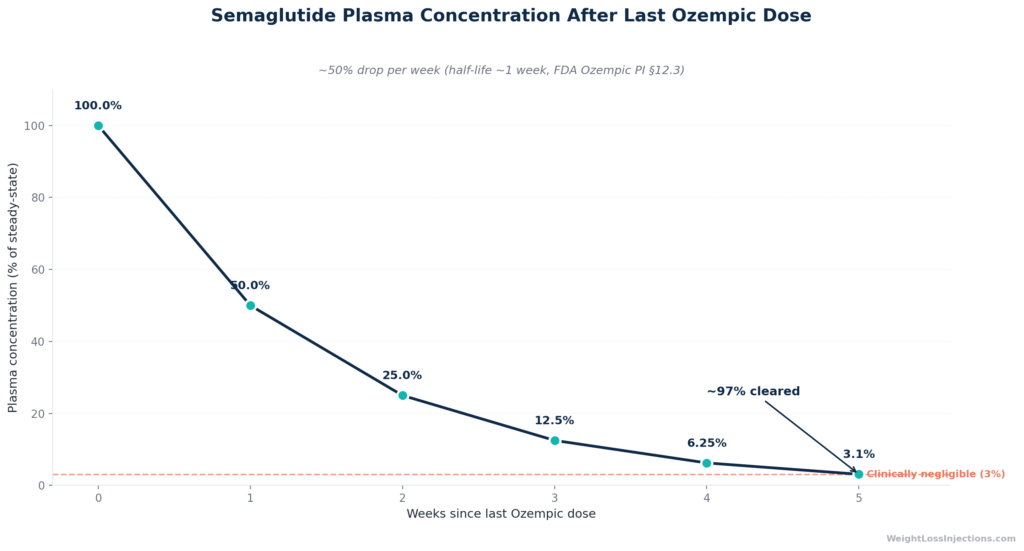
The FDA Ozempic prescribing information, section 12.3 (2025) states it plainly: “With an elimination half-life of approximately 1 week, semaglutide will be present in the circulation for about 5 weeks after the last dose.”
A half-life is the time it takes for plasma concentration to fall by exactly 50%. Because semaglutide’s half-life is ~168 hours (~7 days), the math is straightforward: after each week without a dose, half of what was in your bloodstream at the start of that week is gone. The decay follows predictable first-order kinetics.
Half-life decay table — post-last-dose (at steady-state 1 mg):
| Week After Last Dose | Plasma Level Remaining | Interpretation |
|---|---|---|
| Week 0 (last dose) | ~100% | Steady-state peak |
| Week 1 | ~50% | Appetite suppression still likely noticeable |
| Week 2 | ~25% | Appetite returning; GI effects fading |
| Week 3 | ~12.5% | Residual glycemic benefit limited |
| Week 4 | ~6.25% | Sub-therapeutic for most patients |
| Week 5 | ~3.1% | Clinically negligible; drug effectively cleared |
These figures are derived from the FDA PI §12.3 using standard five-half-life elimination principles, and are consistent with the clinical pharmacokinetics systematic review by Yang & Yang (Drug Design, Development and Therapy, 2024), which found subcutaneous semaglutide half-life ranges of 145–168 hours across multiple steady-state studies.
The underlying protraction mechanism is albumin binding. Per the FDA PI §12.3, “the principal mechanism of protraction resulting in the long half-life of semaglutide is albumin binding” — semaglutide binds >99% to plasma albumin, which dramatically reduces renal clearance and protects the peptide from enzymatic degradation. The apparent clearance is only approximately 0.05 L/hour — extraordinarily slow for a peptide drug.
Reaching Steady-State: The First 4–5 Weeks on Ozempic
Ozempic’s half-life works in both directions: the same slow accumulation that makes it slow to clear also means it takes time to build up. Per the FDA PI §12.3, “steady-state exposure is achieved following 4 to 5 weeks of once-weekly administration.” This is why Ozempic’s titration schedule, starting at 0.25 mg for four weeks before moving to 0.5 mg, is not simply about tolerability. Patients are not at full therapeutic plasma levels during weeks 1–4.
At steady state, mean plasma concentrations are approximately 65 ng/mL at 0.5 mg/week and 123 ng/mL at 1 mg/week, per FDA PI population PK modeling. At the higher 2 mg dose (used in some patients), steady-state concentration reaches approximately 222 ng/mL. Importantly, semaglutide exposure increases in a dose-proportional manner — double the dose, roughly double the plasma level.
Factors Affecting How Long Ozempic Stays in Your System
Renal and Hepatic Impairment: No Clinically Relevant Change
One of the most important, and counterintuitive, findings from the Ozempic PK program is that kidney disease and liver disease do not meaningfully alter how long semaglutide stays in your system. The FDA Ozempic PI §12.3 (2025) is explicit:
“No dose adjustment of OZEMPIC is recommended for patients with renal impairment. In subjects with renal impairment including kidney failure, no clinically relevant change in semaglutide pharmacokinetics (PK) was observed.”
The same finding applies to the liver: “Hepatic impairment does not have any impact on the exposure of semaglutide.”
This matters clinically because many patients on Ozempic have comorbid chronic kidney disease or fatty liver disease. Their worry that these conditions might cause drug accumulation or a prolonged washout is understandable but not supported by the evidence. The reason is mechanistic: semaglutide is not primarily cleared by the kidneys or the liver. It is metabolized across tissues by proteolytic cleavage of the peptide backbone and sequential beta-oxidation of the fatty acid side chain, per FDA PI §12.3. Renal function largely governs how much intact semaglutide exits in urine (approximately 3% of the dose), but that is a minor route, not the rate-limiting step.
The 2024 systematic review in Drug Design, Development and Therapy (Yang & Yang) does note that mean half-life was somewhat longer in severe renal impairment (221 hours) and end-stage renal disease (243 hours) compared to the population-level ~168-hour median, but the FDA’s own population PK analysis, which pooled clinical trial data, did not find a clinically meaningful difference sufficient to require dose adjustment. For most patients with CKD, the practical takeaway is unchanged: expect a five-week washout.
Age, Sex, Race, and Body Weight
Per the FDA PI §12.3 population PK analysis: “age, sex, race, and ethnicity, and renal impairment do not have a clinically meaningful effect on the pharmacokinetics of semaglutide.” An 80-year-old woman does not have meaningfully different clearance than a 30-year-old man.
Body weight is the one exception, but not in the direction most patients assume. Heavier patients actually have lower semaglutide exposure at the same dose, because the volume of distribution scales with body size. The FDA PI Figure 3 shows relative average concentration decreasing with higher body weight (127 kg vs. reference) and increasing at lower body weight (55 kg). However, the PI confirms that doses of 0.5 mg and 1 mg provide adequate systemic exposure across the body weight range of 40 to 198 kg studied in clinical trials, so this does not translate into a meaningful difference in washout time.
Dose: Higher Doses Mean Higher Peak Levels, Not Different Half-Life
The half-life itself is dose-independent, semaglutide’s ~168-hour half-life does not change whether you are on 0.5 mg or 2 mg. What changes is the absolute amount in your system at the time you stop. At 2 mg steady-state (approximately 222 ng/mL), you start from a higher plateau than at 1 mg (approximately 123 ng/mL). The percent decay per week is the same, but absolute plasma levels remain higher at each time point. This is one reason the Wegovy 2.4 mg prescribing information specifies 5–7 weeks of detectable levels (discussed below), compared to the five weeks cited in the Ozempic label.
Detection in Blood, Urine, and Drug Tests
How Long Is Ozempic Detectable?
Semaglutide is detectable in plasma for approximately five weeks after the last Ozempic injection, consistent with the five-half-life elimination principle per FDA PI §12.3. At the analytical level, liquid chromatography–mass spectrometry (LC-MS/MS) can detect semaglutide at concentrations well below the therapeutic range, but clinical plasma assays used in pharmacokinetic studies become unreliable below ~1–2 ng/mL, which corresponds to roughly week 5 at clinical doses.
Urine detection is limited. Per FDA PI §12.3, only approximately 3% of the dose is excreted in urine as intact semaglutide. The vast majority is metabolized intracorporeally and the degradation fragments — not the intact peptide — are what appear in both urine and feces.
Will Ozempic Show Up on a Standard Drug Test?
No. Standard workplace or forensic urine drug screens test for a specific panel of controlled substances (amphetamines, opioids, cannabinoids, cocaine, benzodiazepines, etc.) and do not include GLP-1 receptor agonists. Semaglutide is not a controlled substance, has no abuse potential, and is not on any standard screening panel. A standard urinalysis will not detect it.
Specialized immunoassay or LC-MS confirmation testing could theoretically identify semaglutide if specifically requested (for example, in a research context or therapeutic drug monitoring). But this would be an explicitly ordered test for semaglutide — not something that shows up incidentally on routine screening. If you are preparing for a standard pre-employment or routine health drug screen, Ozempic will not affect your results.
Ozempic vs. Wegovy vs. Rybelsus Clearance Comparison
Ozempic vs. Wegovy vs. Rybelsus: Clearance Compared
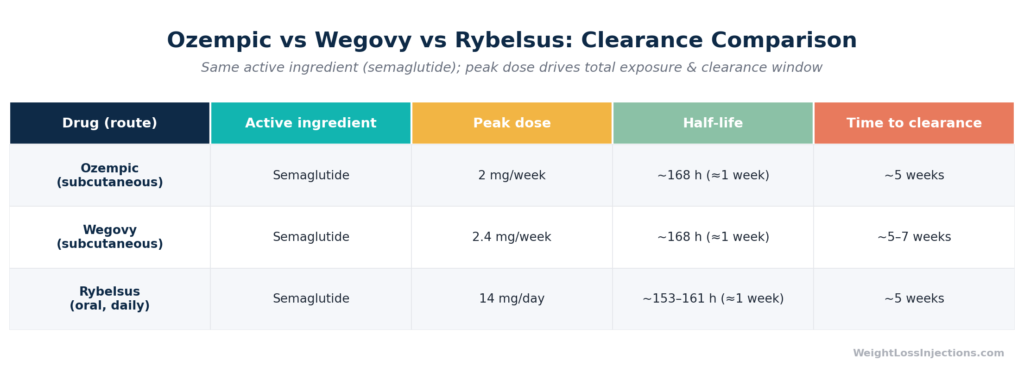
All three semaglutide formulations share the same active molecule, the same albumin-binding protraction mechanism, and the same elimination half-life of approximately one week. But there are meaningful differences in clearance time tied to dosing.
Ozempic (subcutaneous, T2D/CKD): Maximum approved dose is 2 mg/week. Steady-state levels at 1 mg are approximately 123 ng/mL; at 2 mg, approximately 222 ng/mL per FDA Ozempic PI §12.3 (2025). Clearance time: ~5 weeks per the label.
Wegovy (subcutaneous, weight management): Maximum maintenance dose is 2.4 mg/week, with an investigational 7.2 mg dose studied for severe obesity. Steady-state at 2.4 mg is approximately 75 nmol/L (~75 nM), per Wegovy PI §12.3 (Novo Nordisk). Because the starting plateau is higher, the Wegovy PI explicitly states that semaglutide “will be present in the circulation for about 5 to 7 weeks after the last injectable dose of 2.4 mg or 7.2 mg” — slightly longer than the five weeks cited for Ozempic’s lower doses. The Yang & Yang systematic review (DDDT, 2024) confirmed semaglutide 2.4 mg has a half-life of 145–165 hours, statistically identical to the 0.5 and 1 mg doses.
Rybelsus (oral, T2D): Oral bioavailability is approximately 1% under standard fasting conditions per the DDDT systematic review (Yang & Yang, 2024) (compared to 89% for subcutaneous Ozempic per FDA PI §12.3). Despite this dramatically lower systemic bioavailability, once oral semaglutide is absorbed, its elimination half-life is essentially the same — approximately 153–161 hours at steady state, per Yang & Yang, DDDT 2024. Steady-state concentrations are lower than subcutaneous administration, so Rybelsus users stopping the medication reach clinically negligible levels in approximately five weeks.
The key insight: route of administration changes how much drug gets into the body, but not how long it takes to leave once it’s there.
What Happens After Your Last Dose
Therapeutic Effects and Side Effects Fade Gradually
Because semaglutide does not disappear abruptly, its effects after stopping follow the same five-week decay curve as the plasma levels. In the first one to two weeks, most patients notice progressively increasing appetite, the return of “food noise”, as GLP-1 receptor activation wanes. Gastric emptying returns toward baseline, so patients who relied on slowed emptying for satiety may feel hungrier sooner after meals.
For type 2 diabetes patients, blood glucose will begin rising as GLP-1-driven insulin secretion and glucagon suppression diminish. The Mayo Clinic semaglutide medication guide recommends increased glucose monitoring in the weeks following discontinuation.
GI side effects, nausea, diarrhea, constipation, typically resolve within two to three weeks of stopping, tracking closely with the plasma decay curve. Some patients actually experience a pleasant relief from GI effects by week two post-stop.
What about rebound weight regain? Clinical trial data are sobering. The SUSTAIN and STEP extension trials demonstrated that most patients regain a substantial proportion of lost weight within one to two years of stopping semaglutide, even without any change in diet or activity. This is not a withdrawal syndrome, there are no physical withdrawal symptoms from stopping Ozempic per GoodRx pharmacist guidance. The weight regain reflects the underlying chronic nature of obesity and diabetes; the drug was managing, not curing, a physiologic condition.
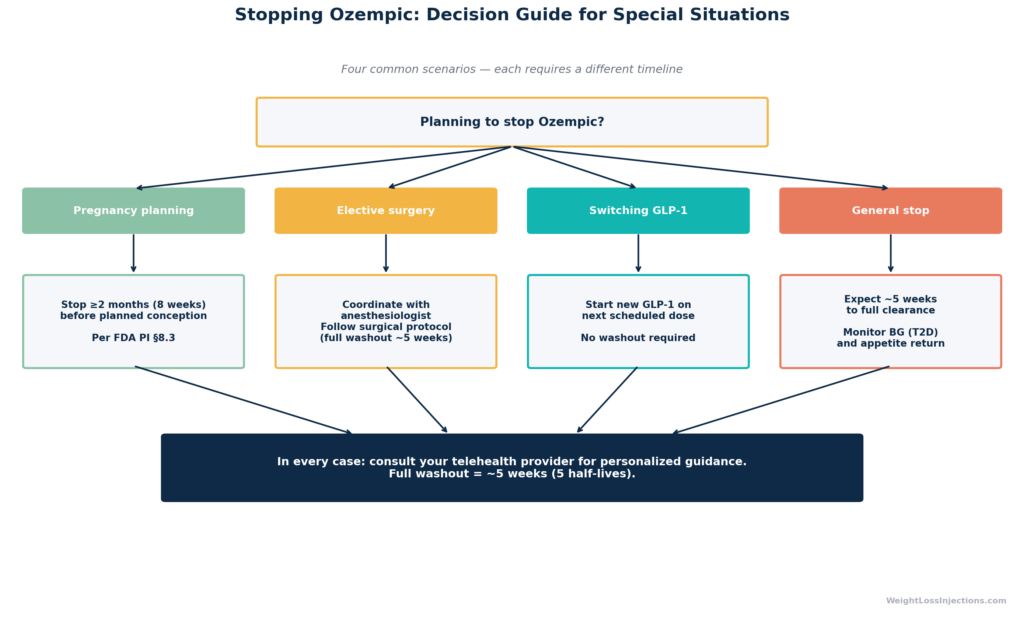
Pregnancy: Stop at Least Two Months Before Planned Conception
The FDA Ozempic PI §8.1 and §8.3 (2025) contains a specific pregnancy guidance statement: “Discontinue OZEMPIC in women at least 2 months before a planned pregnancy due to the long washout period for semaglutide.”
This two-month (approximately eight weeks) buffer provides a margin beyond the five-week clinical clearance window. Semaglutide’s safety in human pregnancy has not been established, and animal studies showed embryofetal toxicity at clinically relevant exposures. The European Medicines Agency Rybelsus product information uses identical language, recommending discontinuation at least two months before planned pregnancy, applicable to all semaglutide formulations.
This two-month window applies regardless of which semaglutide product you are using. If you are planning a pregnancy, discuss the timing with your prescribing provider well in advance so your last injection can be scheduled accordingly.
Switching GLP-1s: No Mandatory Washout, But Timing Matters
One of the most practically important questions for telehealth patients is: when can you switch from Ozempic to another GLP-1 agonist (or vice versa) without overlap or gap?
There is no contraindication to starting a new GLP-1 immediately after the last Ozempic dose, the drugs work through the same receptor, so there is no interaction risk. In practice, most providers schedule the first dose of the new medication at the same time as the next scheduled Ozempic dose would have been. This minimizes both overlap (additive GI effects) and the gap in coverage.
When switching to a non-GLP-1 medication (for example, Mounjaro/tirzepatide, which is a dual GLP-1/GIP agonist), the same approach generally applies. Per WeightLossInjections.com’s telehealth guidance on stopping GLP-1 meds safely, your provider can also tailor the transition timing if you need a clean washout, for surgical procedures, for example.
For surgical procedures: Anesthesiology guidance has evolved on this topic. Concern about aspiration risk from gastroparesis has led some societies to recommend pausing GLP-1 agonists before elective surgery. With semaglutide’s five-week washout, a provider-directed temporary hold gives anesthesiologists adequate lead time, but follow your surgical team’s specific protocol.
Flowchart: Special Situation Decision Guide for Stopping Ozempic
When to Consult Your Telehealth Provider
While Ozempic’s elimination timeline is predictable at the population level, your individual context may warrant a customized plan. Consider contacting your WeightLossInjections.com [service detail] provider for guidance if you are:
- Planning a pregnancy — Calculate your last injection date based on two months before your target conception window, not just five weeks. This is not a number to estimate; confirm the timing with your provider.
- Scheduled for surgery — Ask specifically about your institution’s GLP-1 hold policy. Some anesthesiologists now request holds of one to two weeks; others, particularly for short outpatient procedures, may not. Your provider can draft a brief note for your surgical team.
- Switching GLP-1 medications — If you are moving from Ozempic to Wegovy, Mounjaro, or another agent, a simple handoff plan prevents gap-and-restart cycles that can trigger unnecessary GI side effects from re-titration.
- Experiencing prolonged side effects — GI symptoms persisting more than five to six weeks after your last dose, or symptoms that worsen rather than improve post-stop, warrant evaluation.
- A dose was missed before stopping — If your last “dose” was actually a missed-dose situation from several weeks ago, your clearance calculation should be referenced to the last actual injection.
WeightLossInjections.com offers telehealth consultations at [$X/month] that include access to licensed prescribers who can review your individual medical history and provide personalized guidance. Our network supports patients through medication starts, transitions, and planned stops.
Our take at WeightLossInjections.com: The five-week elimination timeline is a framework, not a guarantee. It reflects population averages from well-controlled PK studies. Individual variation exists — a 55 kg patient may clear the drug somewhat faster than a 127 kg patient at peak steady-state (due to the body weight–exposure relationship noted in the FDA PI). And the practical endpoints that matter to patients — when will my appetite come back? when will my blood sugar start rising? — track with plasma levels but are subject to individual metabolic variation. The data give you a map; your provider helps you navigate the terrain.
Frequently Asked Questions
How many weeks until Ozempic is fully out of my system?
Using the standard pharmacokinetic measure of five half-lives, Ozempic is effectively eliminated from your system in approximately five weeks after your last dose. The FDA Ozempic prescribing information §12.3 (2025) states this explicitly: “semaglutide will be present in the circulation for about 5 weeks after the last dose.” At this point, less than 3.1% of peak steady-state concentration remains — a level considered clinically negligible. For Wegovy at its maximum dose of 2.4 mg, the Wegovy PI (Novo Nordisk) extends this estimate to 5–7 weeks, reflecting the higher plasma levels from which clearance begins. “Fully out” by analytical detection is a somewhat different bar: LC-MS/MS can detect trace amounts beyond five weeks, but these concentrations have no therapeutic or physiologic significance.
Does kidney or liver disease change Ozempic clearance time?
For most patients, no. The FDA Ozempic PI §12.3 (2025) explicitly states that no clinically relevant change in semaglutide pharmacokinetics was observed in studies of patients with mild, moderate, severe renal impairment, or kidney failure — and the same applies to hepatic impairment across all severity levels. This finding makes mechanistic sense: only approximately 3% of semaglutide exits via urine as intact drug, so even severely impaired kidneys don’t cause meaningful accumulation. However, the Yang & Yang systematic review (DDDT, 2024) did find statistically longer half-lives in severe renal impairment (221 hours) and end-stage renal disease (243 hours) vs. normal renal function (~168 hours). The FDA concluded these differences were not large enough to warrant dose adjustment or changed prescribing, but patients with ESRD should still discuss clearance expectations with their nephrologist. The standard guidance remains: no dose adjustment required, and expect approximately five weeks for clearance.
Will Ozempic show up on a drug test?
No — not on any standard workplace, pre-employment, or clinical urinalysis drug screen. These tests use immunoassay panels targeting controlled substances: opioids, amphetamines, cocaine, cannabinoids, benzodiazepines, and related compounds. Semaglutide is not a controlled substance, does not share structural or chemical similarity with tested compounds, and is not screened for. As noted above, only approximately 3% of the dose appears as intact semaglutide in urine, and specialized research-grade assays (LC-MS/MS) would be required to detect it. In a clinical or research setting where a provider specifically orders semaglutide plasma levels, for therapeutic drug monitoring or compliance checking, it would be detectable for approximately five weeks. But routine drug screens do not test for it and will not flag it.
How long after stopping Ozempic can I get pregnant?
The FDA Ozempic prescribing information §8.3 (2025) provides a clear minimum: “Discontinue OZEMPIC in women at least 2 months before a planned pregnancy due to the long washout period for semaglutide.” Two months, approximately eight weeks, provides a conservative margin beyond the five-week clinical clearance window. This recommendation is based on animal reproductive toxicology data showing embryofetal effects at doses comparable to human therapeutic exposures; formal human safety data in pregnancy are not available. The same two-month pre-conception washout recommendation appears in the European Medicines Agency Rybelsus product information and applies to all semaglutide formulations (Ozempic, Wegovy, Rybelsus). If you are using Ozempic for type 2 diabetes management and plan to become pregnant, work with your endocrinologist to transition to a diabetes medication with an established pregnancy safety profile (such as insulin) well before your target conception window.
Can I speed up Ozempic leaving my body?
There is no established method to accelerate semaglutide clearance. This is because the rate-limiting step is not renal filtration or hepatic metabolism, processes that could theoretically be modified, but rather semaglutide’s tight albumin binding and slow proteolytic degradation across tissues. You cannot meaningfully dialyze it out (albumin-bound drugs are not dialyzable), force its excretion, or speed up the enzymatic metabolism. Per the FDA PI, there is no antidote to semaglutide exposure, and no specific reversal agent exists. This is clinically relevant in overdose contexts, if someone receives an accidental overdose, prolonged observation and symptomatic management are required, as semaglutide cannot be rapidly removed from the system. The DrugBank semaglutide monograph echoes this: “Prolonged observation and treatment may be required, as the half-life of this drug is about one week. There is no antidote to an overdose with semaglutide.” For patients who simply want the drug gone faster (due to side effects, surgery, or a medical procedure), the practical guidance is planning: stop early enough that the five-week washout completes before your target date.
WeightLossInjections.com Editorial Note: This article reflects pharmacokinetic data published in the FDA-approved prescribing information and peer-reviewed literature as of April 2026. Semaglutide PK has been studied extensively across populations, and the five-week elimination framework is well-validated. That said, clinical decisions around stopping, holding, or switching Ozempic should always be made with your prescribing provider, not based on population averages alone. WeightLossInjections.com’s telehealth platform connects patients with licensed prescribers for exactly this kind of individualized guidance at [$X/month].

