SUSTAIN-2 Trial: Semaglutide Added to Metformin vs. Sitagliptin — Key Outcomes
Yes. Taking metformin and Ozempic (semaglutide) together is a well-established, guideline-endorsed combination for adults with type 2 diabetes (T2D). The ADA 2026 Standards of Medical Care in Diabetes position metformin as the standard first-line agent and GLP-1 receptor agonists, including Ozempic, as a preferred add-on when A1c targets are not met, particularly in patients with cardiovascular risk.
A dedicated pharmacokinetic (PK) study confirms no clinically significant drug interaction. The main management consideration is stacked GI side effects: both drugs affect the gut, and dose titration strategy matters.
Safety notice: This article is for educational purposes only and does not constitute medical advice. Always consult a licensed healthcare provider before starting, combining, or adjusting medications for type 2 diabetes or weight management.
Can You Take Metformin and Ozempic Together Safely?
The answer is yes, and it is not a borderline call. Combining metformin and semaglutide (Ozempic) is explicitly addressed in the FDA Ozempic Prescribing Information (NDA 209637), which documents multiple phase 3 clinical trials conducted on top of background metformin therapy, including the 56-week SUSTAIN-2 trial, the primary evidence base for Ozempic’s approval in combination therapy. The label lists no drug-drug interaction concern and confirms that no dose adjustment of either agent is required.
Mechanistically, there is no pharmacokinetic reason for concern. A rigorous Phase 1 PK interaction study (Hausner et al., 2017, PMC5648738) enrolled 22 healthy subjects to measure metformin exposure (500 mg twice daily) before and during steady-state subcutaneous semaglutide 1.0 mg. The results showed an AUC ratio of 1.03 (90% CI: 0.96–1.11) and a Cmax ratio of 0.90 (90% CI: 0.83–0.98), both within the pre-specified bioequivalence interval of 0.80–1.25. The modest 10% reduction in metformin peak concentration reflects semaglutide’s gastric emptying delay but has no clinically meaningful impact on overall drug exposure. The study concluded: no clinically significant pharmacokinetic interaction; no dose adjustment required.
When Providers Prescribe This Combination
In current clinical practice, this combination most commonly arises in one of two scenarios:
- Metformin first, Ozempic added: A patient with T2D starts metformin as first-line therapy per standard of care. If A1c remains above target (typically above 7.0%), the ADA 2026 Standards of Medical Care in Diabetes recommend adding a GLP-1 receptor agonist with demonstrated cardiovascular benefit — semaglutide among them — particularly when the patient has established atherosclerotic cardiovascular disease (ASCVD) or high cardiovascular risk.
- Ozempic prescribed, metformin continued: A patient already stabilized on metformin is transitioned to or initiated on Ozempic for additional glycemic control, weight reduction, or cardiovascular risk reduction, without discontinuing the underlying metformin.
In both cases, continuing metformin is not only safe but actively recommended. Metformin provides basal glucose control through hepatic mechanisms that are entirely complementary to Ozempic’s GLP-1 receptor-mediated actions.
Important regulatory note: Ozempic (semaglutide 0.5 mg, 1 mg, 2 mg) is FDA-approved for glycemic control in adults with type 2 diabetes, with a separate cardiovascular indication (reducing MACE risk). It is not FDA-approved as a standalone weight-loss medication. That indication belongs to Wegovy (semaglutide 2.4 mg, approved June 4, 2021, NDA 215256). If your provider is combining metformin and semaglutide, it should be in the context of T2D management, not off-label weight loss.
How Metformin and Ozempic Work Together
Mechanisms of Action
Metformin and semaglutide reach glucose control through entirely different pathways, which is precisely why they complement each other.
Metformin (a biguanide) works primarily by suppressing hepatic glucose production, reducing the amount of glucose the liver releases into the bloodstream between meals and overnight. Secondarily, it modestly improves peripheral insulin sensitivity in skeletal muscle and reduces intestinal glucose absorption. It does not stimulate insulin secretion, which means it carries a very low risk of hypoglycemia as monotherapy. The FDA metformin prescribing information describes it as addressing the insulin resistance component of T2D pathophysiology.
Ozempic (semaglutide) is a GLP-1 receptor agonist, a synthetic analog of glucagon-like peptide-1 that is structurally modified to resist degradation and allow once-weekly dosing. Its mechanism is multi-pronged: it augments glucose-stimulated insulin secretion from pancreatic beta cells, suppresses glucagon release from alpha cells (reducing hepatic glucose output), slows gastric emptying (blunting post-meal glucose spikes), and acts on hypothalamic satiety centers to reduce appetite and caloric intake. Per the FDA Ozempic Prescribing Information, semaglutide’s insulin secretion effect is glucose-dependent, it diminishes as blood glucose normalizes, substantially limiting hypoglycemia risk when used without sulfonylureas or insulin.
Synergistic Benefits
The combination addresses T2D from multiple angles simultaneously:
- Metformin targets the liver (fasting glucose, insulin resistance)
- Ozempic targets the pancreas (post-meal glucose), stomach (gastric emptying), and brain (appetite/satiety)
- Together they produce greater fasting blood glucose reduction, post-meal glucose attenuation, A1c lowering, and weight loss than either agent alone
A 2024 meta-analysis published in the American Journal of Translational Research (AJTR, PMC11384358) pooled 10 randomized controlled trials (962 patients with T2D who were overweight or obese) comparing semaglutide plus metformin against metformin alone. The combination arm demonstrated statistically significant superiority across every measured metabolic outcome: HbA1c (SMD −1.13, 95% CI: −1.85 to −0.42, p<0.001), fasting blood glucose (SMD −0.94, p<0.001), post-meal 2-hour glucose (SMD −0.97, p<0.0001), BMI (SMD −1.08, p<0.0001), HOMA-IR insulin resistance index (SMD −0.92, p<0.0001), and lipid panels including total cholesterol (SMD −1.03, p<0.001) and triglycerides (SMD −0.96, p<0.001). Adverse event rates were not significantly different between arms.
Clinical Evidence: The SUSTAIN-2 Trial
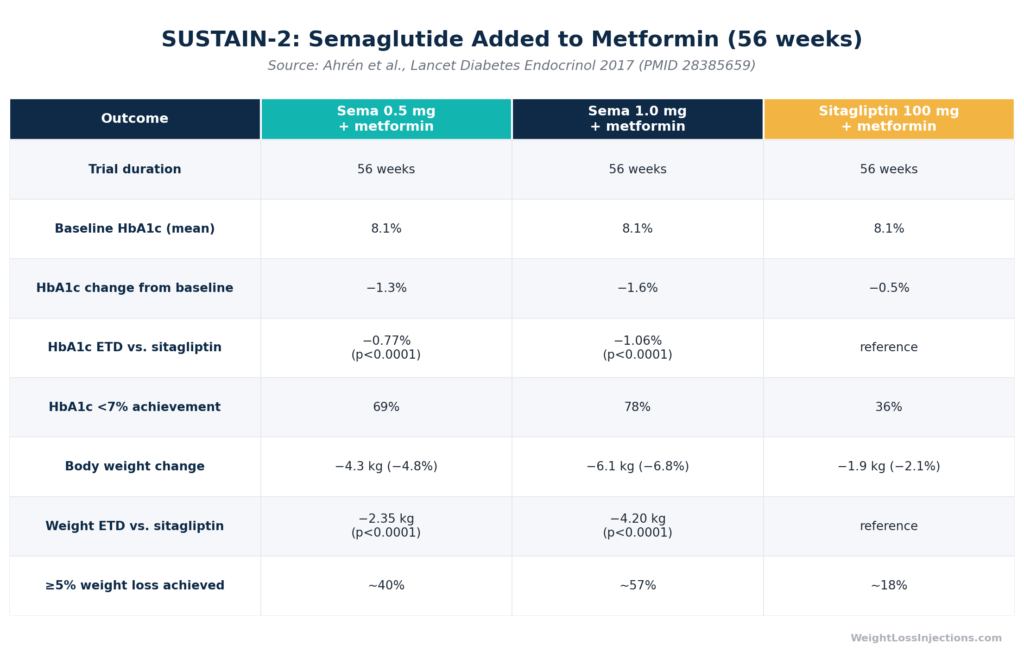
The most direct evidence for the metformin-plus-semaglutide combination comes from SUSTAIN-2 (Ahrén et al., Lancet Diabetes & Endocrinology, 2017; PMID 28385659), the global phase 3a trial that formed the core of Ozempic’s FDA approval for combination therapy.
Design: 56-week, double-blind, double-dummy, active-controlled, parallel-group multinational trial across 128 sites in 18 countries. A total of 1,231 adults with T2D and A1c of 7.0–10.5% on stable background metformin (or metformin plus thiazolidinedione) were randomized to once-weekly subcutaneous semaglutide 0.5 mg, semaglutide 1.0 mg, or once-daily oral sitagliptin 100 mg.
Key results (week 56):
- HbA1c reduction from baseline of 8.1%: −1.3% (semaglutide 0.5 mg), −1.6% (semaglutide 1.0 mg), vs. −0.5% (sitagliptin); estimated treatment differences of −0.77% and −1.06%, both p<0.0001
- A1c target of <7.0% achieved: 69% (0.5 mg), 78% (1.0 mg) vs. 36% (sitagliptin)
- Body weight change from baseline of ~89.5 kg: −4.3 kg (0.5 mg), −6.1 kg (1.0 mg) vs. −1.9 kg (sitagliptin); ETDs of −2.35 kg and −4.20 kg, both p<0.0001
SUSTAIN-2 is the landmark data point establishing that adding Ozempic to metformin produces A1c reductions roughly 2–3× greater than adding sitagliptin, with concurrent and clinically meaningful weight loss. The trial was conducted on a metformin background, meaning every benefit demonstrated was the increment above and beyond what metformin alone was already delivering.
The FDA Ozempic prescribing information also cites SUSTAIN-4 (semaglutide vs. insulin glargine, largely on metformin background) and SUSTAIN-8 (semaglutide vs. canagliflozin, metformin background), all showing consistent HbA1c and weight reductions in the 1.2–1.5% and 3.5–6 kg range.
Side Effects: Where Metformin and Ozempic Overlap, and Where They Diverge
Side-Effect Profile: Metformin vs. Ozempic — Overlap and Distinct Risks
Overlapping GI Effects — and How to Manage Them
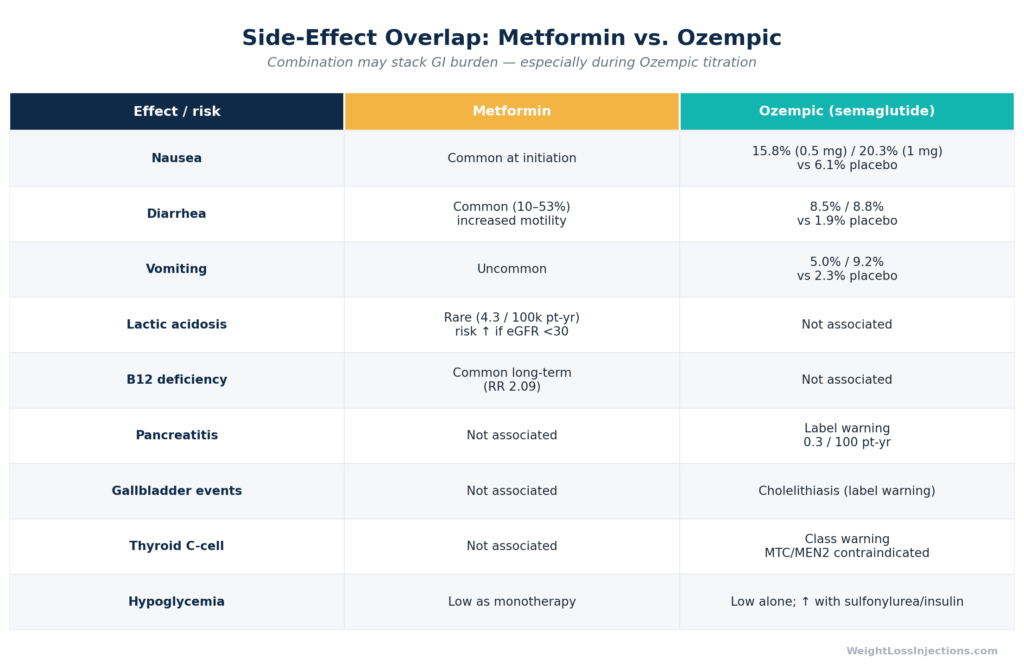
The most clinically relevant concern when combining metformin and Ozempic is stacked gastrointestinal side effects. Both drugs cause GI symptoms, primarily through different mechanisms, but the practical experience of nausea, diarrhea, and GI discomfort can be additive during Ozempic initiation.
From the FDA Ozempic Prescribing Information, GI adverse reactions in pooled placebo-controlled trials occurred in 32.7% (0.5 mg) and 36.4% (1 mg) of semaglutide-treated patients vs. 15.3% with placebo, with the majority occurring during dose escalation. Nausea rates were 15.8–20.3% vs. 6.1% placebo. Diarrhea rates were 8.5–8.8% vs. 1.9% placebo.
Metformin’s GI effects, bloating, diarrhea, nausea, are well-characterized and affect up to 25–30% of patients at standard doses, per the FDA metformin labeling. They are dose-dependent and most pronounced at initiation.
Practical dose-titration strategy to minimize combined GI effects:
- Start Ozempic at the lowest dose (0.25 mg/week × 4 weeks) as directed in the prescribing information — this is a run-in period, not a therapeutic dose, specifically designed to allow GI tolerance to develop before dose escalation.
- Take metformin with food. This consistently reduces GI side effects and is standard prescribing practice per the FDA metformin PI. If not already doing so, patients switching from immediate-release to extended-release (ER) metformin often experience substantially fewer GI symptoms.
- Do not escalate both drugs simultaneously. If you are starting Ozempic on top of metformin, keep the metformin dose stable during Ozempic titration, then address any metformin dose changes in a separate step.
- Eat smaller, lower-fat meals. High-fat meals amplify both Ozempic-induced nausea and metformin GI effects. GI symptoms on this combination tend to peak in the first 4–8 weeks and attenuate substantially as the body adjusts.
- Stay hydrated. Dehydration concentrates metformin and increases lactic acidosis risk at any eGFR.
Metformin-Specific Risks
Lactic acidosis is the most serious, and most misunderstood, risk associated with metformin. It is genuinely rare: estimated at approximately 4.3 cases per 100,000 patient-years among metformin users. A large retrospective cohort study published in JAMA Internal Medicine (Lazarus et al., 2018; PMC6145716) — covering more than 157,000 patients across two cohorts, found that metformin use was not associated with increased acidosis risk at eGFR ≥30 mL/min/1.73 m². The risk elevated meaningfully only at eGFR <30 (adjusted HR 2.07, 95% CI: 1.33–3.22). The FDA metformin label reflects this: metformin is contraindicated at eGFR <30, not recommended for new initiation at eGFR <45, and requires renal monitoring during ongoing therapy.
Vitamin B12 deficiency is a more common and underappreciated consequence of long-term metformin use. A meta-analysis of 31 studies found that metformin users had a relative risk of 2.09 for vitamin B12 deficiency (95% CI: 1.49–2.93) compared with non-users, with average B12 levels approximately 63.70 pM lower. The risk is dose- and duration-dependent: doses ≥2,000 mg/day and duration ≥4 years carry significantly elevated risk, per data reviewed in the World Journal of Diabetes. Clinically, B12 deficiency can manifest as macrocytic anemia or peripheral neuropathy — symptoms that can be mistaken for diabetic neuropathy.
The ADA 2026 Standards of Medical Care in Diabetes recommend periodic B12 monitoring in long-term metformin users, with annual B12 testing suggested for patients taking metformin for 4+ years or with other risk factors (older age, higher doses, PPI use, dietary restrictions). The FDA metformin label recommends measuring B12 levels every 2–3 years in all patients on metformin.
Ozempic-Specific Risks
Pancreatitis: The FDA Ozempic Prescribing Information lists pancreatitis as a warning and precaution. Incidence in clinical trials was 0.3 cases per 100 patient-years on semaglutide vs. 0.2 on comparators, a small numerical difference not established as statistically significant. Ozempic has not been studied in patients with a prior history of pancreatitis; if pancreatitis is confirmed on therapy, Ozempic should not be restarted.
Gallbladder events: Cholelithiasis (gallstones) and cholecystitis have been reported with semaglutide, consistent with the GLP-1 class effect of reducing gallbladder motility. The FDA PI lists these as adverse reactions to be aware of; patients with pre-existing gallstone disease should discuss risks with their prescriber.
Thyroid C-cell tumors: Semaglutide carries a boxed warning for risk of thyroid C-cell tumors, based on rodent carcinogenicity studies showing dose-dependent thyroid C-cell tumors at clinically relevant exposures. Whether this risk translates to humans is unknown. Ozempic is contraindicated in patients with a personal or family history of medullary thyroid carcinoma (MTC) or Multiple Endocrine Neoplasia syndrome type 2 (MEN 2).
Hypoglycemia: Ozempic alone, or Ozempic plus metformin, carries low hypoglycemia risk because both agents have glucose-dependent mechanisms. Hypoglycemia risk increases substantially when a sulfonylurea or insulin is added to the combination.
Dosage and Titration in Combination
Metformin Dosing
Per the FDA metformin prescribing information, standard adult dosing is:
- Immediate-release: Start at 500 mg twice daily or 850 mg once daily with meals, titrate by 500 mg weekly or 850 mg every 2 weeks to target. Maximum effective dose: 2,550 mg/day; maximum tolerated dose for many patients is 1,500–2,000 mg/day.
- Extended-release (ER): Start at 500–1,000 mg/day with evening meal; titrate weekly. Maximum: 2,000–2,500 mg/day depending on formulation. ER is preferred for GI tolerability.
Renal dose adjustment is required: initiate at eGFR ≥45; reassess at eGFR 30–45; contraindicated at eGFR <30.
Ozempic Dosing When Added to Metformin
Per the FDA Ozempic Prescribing Information:
- Week 1–4: 0.25 mg subcutaneous injection once weekly (tolerability run-in, not therapeutic)
- Week 5–8+: 0.5 mg once weekly (first therapeutic dose)
- If additional glycemic control needed after ≥4 weeks at 0.5 mg: Increase to 1.0 mg once weekly
- If further control needed after ≥4 weeks at 1.0 mg: Increase to 2.0 mg once weekly (approved January 2022)
Ozempic requires no renal dose adjustment, per the FDA PI, though patients with CKD should discuss the appropriate combination of diabetes medications with their nephrologist or endocrinologist.
Who Should Not Use This Combination, or Use It With Caution
Absolute Contraindications for Metformin
- eGFR <30 mL/min/1.73 m² (per FDA metformin PI)
- Acute or unstable conditions predisposing to lactic acidosis: sepsis, dehydration, acute MI, respiratory failure, hepatic insufficiency, acute alcohol intoxication
- Planned iodinated contrast procedure (hold metformin perioperatively if eGFR <60)
Absolute Contraindications for Ozempic
- Personal or family history of medullary thyroid carcinoma (MTC) or MEN 2 (per FDA Ozempic PI)
- Serious hypersensitivity to semaglutide or any excipient (anaphylaxis, angioedema)
- Use in combination with another GLP-1 receptor agonist or a DPP-4 inhibitor (no additive benefit; the ADA 2026 Standards explicitly state never to combine GLP-1 RA and DPP-4 inhibitor)
Use With Caution
- Prior pancreatitis history: Ozempic has not been studied in this population; careful risk-benefit evaluation required
- Gallstone disease: Consider surgical evaluation before starting Ozempic if gallstones are symptomatic
- eGFR 30–44: Metformin use requires individualized risk-benefit assessment and more frequent renal monitoring
- Pregnancy: Neither drug has established safety data in pregnancy; Ozempic should be discontinued at least 2 months before a planned pregnancy per FDA PI guidance
Clinical Decision Algorithm: When to Add Ozempic to Metformin
ADA 2026 T2D Treatment Algorithm — When to Add Ozempic to Metformin
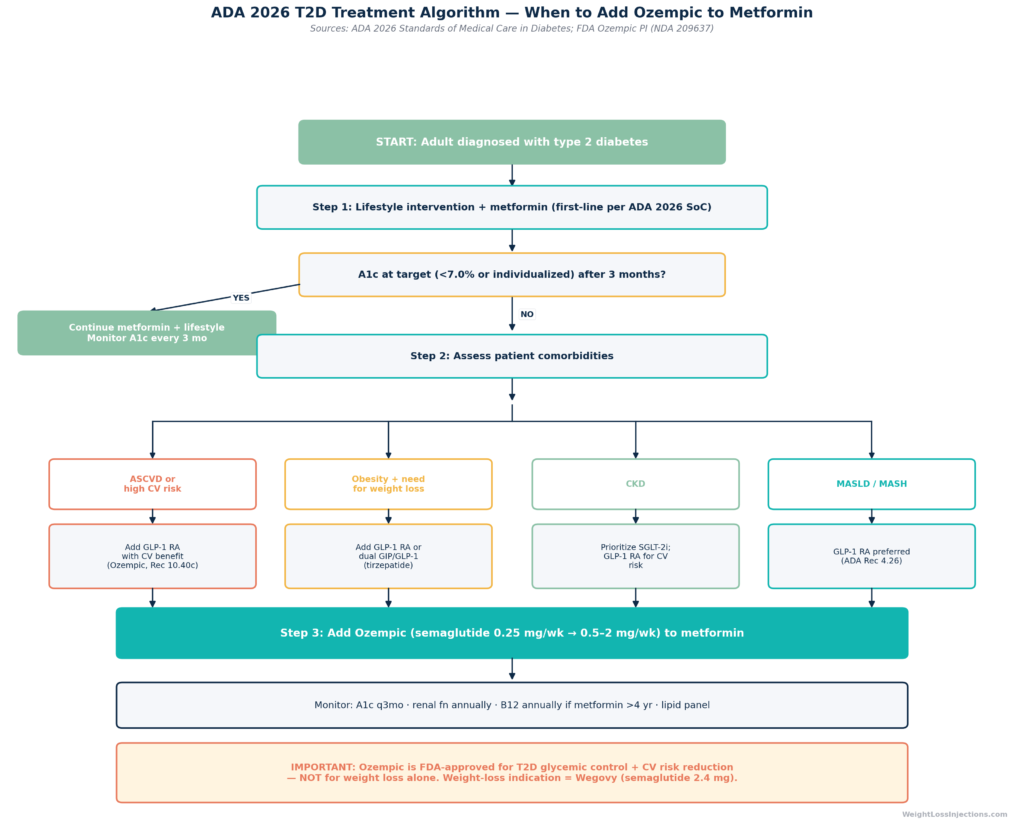
The ADA 2026 Standards of Medical Care in Diabetes use a comorbidity-driven algorithm rather than a simple step-therapy ladder. The key updated recommendations relevant to the metformin-plus-semaglutide combination include:
- Recommendation 10.40c: In adults with T2D and established ASCVD or high cardiovascular risk, a GLP-1 receptor agonist with demonstrated CV benefit is a preferred add-on to metformin.
- Recommendation 4.26: In adults with T2D, MASLD (metabolic-associated steatotic liver disease), and overweight or obesity, a GLP-1 RA is recommended for obesity management with potential MASH benefit.
- General standard: Metformin remains central to T2D pharmacotherapy. Treatment plans should incorporate healthy behaviors, diabetes self-management education, and attention to social determinants of health.
The 2026 guidelines from DrAlo.net reviewing ADA vs. NICE note that the ADA does not mandate a universal SGLT-2 inhibitor at first line (unlike NICE), instead taking a comorbidity-driven approach — meaning a GLP-1 RA like semaglutide may be the preferred second agent in many clinical scenarios, added on top of metformin.
Monitoring Schedule When Combining Both Drugs
| Parameter | Frequency | Rationale |
|---|---|---|
| HbA1c | Every 3 months until stable, then every 6 months | Assess treatment efficacy |
| eGFR / serum creatinine | At least annually; every 3–6 months if eGFR 30–60 | Metformin renal safety |
| Vitamin B12 | Every 2–3 years (FDA label); annually if on metformin ≥4 years or high risk (ADA 2026) | Metformin-induced B12 depletion |
| Fasting lipid panel | Annually | Ozempic adds lipid benefit; baseline tracking |
| Abdominal symptoms | At every visit (first 6 months) | Early detection of pancreatitis vs. GI side effects |
Metformin and Ozempic for Weight Loss Off-Label
This section requires a precise clinical distinction.
For people with type 2 diabetes: The combination of metformin and Ozempic produces meaningful weight loss as a secondary benefit of glucose control. The SUSTAIN-2 trial demonstrated −4.3 to −6.1 kg of body weight loss over 56 weeks, on top of the −1.9 kg from metformin/sitagliptin. The 2024 AJTR meta-analysis showed a BMI reduction SMD of −1.08 vs. metformin alone. These are averages across T2D trial populations; individual results vary.
For people without type 2 diabetes who want weight loss only: Ozempic is not approved for this indication, and prescribing it alongside metformin for weight loss in non-diabetic individuals is off-label use. The approved weight-loss medication is Wegovy (semaglutide 2.4 mg), which in the STEP 1 trial produced 14.9% average body weight loss at 68 weeks in people with obesity without diabetes. Metformin is also used off-label for weight management in non-diabetic individuals with insulin resistance, including PCOS, but these are clinical judgment calls requiring a prescriber consultation, not something to self-initiate.
If your interest is weight loss and you have T2D, the metformin-plus-Ozempic combination may legitimately serve both goals. If you do not have T2D, the clinical picture is different and requires a more individualized evaluation.
Our take at WeightLossInjections.com: The metformin-plus-Ozempic combination is not a gray area, it is standard of care, backed by robust phase 3 trial data and embedded in both FDA labeling and the ADA 2026 algorithm. Where patients and providers alike sometimes stumble is in managing the practical reality of two drugs that both affect the GI tract. The solution is not to avoid the combination, but to titrate deliberately: stabilize on extended-release metformin before adding Ozempic, start Ozempic at 0.25 mg and hold there for a full four weeks, adjust meal composition, and set realistic expectations that the first 8 weeks will include some GI adjustment period. The other management point that often gets missed is B12 monitoring, a simple annual blood draw that can prevent irreversible neuropathy in patients on long-term metformin. At WeightLossInjections.com, our telehealth consultations at [$X/month] include a structured review of [service detail], B12 and renal lab tracking, and individualized dose-titration planning so you are not navigating these decisions alone.
Frequently Asked Questions
Is it safe to take metformin and Ozempic together?
Yes. The combination is supported by the FDA Ozempic prescribing information, a dedicated pharmacokinetic interaction study confirming no clinically significant interaction, and the ADA 2026 Standards of Medical Care in Diabetes, which recommends exactly this combination for adults with type 2 diabetes who need additional glycemic control, weight management, or cardiovascular risk reduction beyond what metformin alone provides. The primary management consideration is overlapping GI side effects, which are handled with careful dose titration and extended-release metformin formulations.
What are the side effects of taking metformin and Ozempic together?
The most common side effects are gastrointestinal: nausea, diarrhea, vomiting, and abdominal discomfort. Both drugs cause GI effects by different mechanisms, and the combination can stack these effects, particularly during Ozempic dose escalation. Per the FDA Ozempic PI, nausea occurred in 15.8–20.3% of semaglutide patients vs. 6.1% with placebo. Rare but serious risks unique to each drug include: lactic acidosis (metformin, risk concentrated at eGFR <30 per Lazarus et al.), B12 deficiency (metformin, long-term use), pancreatitis (Ozempic, 0.3 cases/100 patient-years), and gallbladder events (Ozempic). The thyroid C-cell tumor boxed warning for Ozempic makes it contraindicated in patients with personal or family history of MTC or MEN 2.
How much weight can you lose on metformin and Ozempic?
Average weight loss in the SUSTAIN-2 trial, which studied semaglutide added to metformin, was 4.3 kg (0.5 mg dose) to 6.1 kg (1.0 mg dose) over 56 weeks, compared with 1.9 kg for sitagliptin added to metformin. The 2024 AJTR meta-analysis found a statistically significant BMI reduction with semaglutide-plus-metformin vs. metformin alone (SMD −1.08, p<0.0001). Individual results depend on baseline weight, diet, activity level, dose, and duration of therapy. These figures are from T2D trial populations; Ozempic is not FDA-approved for weight loss alone (that indication belongs to Wegovy at the 2.4 mg dose).
Can you take metformin and Ozempic if you don’t have diabetes?
Ozempic is FDA-approved for type 2 diabetes, not for weight loss in non-diabetic individuals. Using it off-label for weight loss without diabetes is not addressed in the approved prescribing information. The FDA-approved weight-loss version of semaglutide is Wegovy (2.4 mg). Metformin is also used off-label in non-diabetic conditions like prediabetes and PCOS, where evidence supports some metabolic benefit, but these are clinical judgment calls. Any off-label combination requires a prescribing provider who can evaluate individual benefits and risks.
Does insurance cover Ozempic with metformin?
Coverage depends heavily on your plan and diagnosis code. For patients with a T2D diagnosis, Ozempic is widely covered by commercial insurers, though prior authorization requirements and formulary tier placement vary significantly. Per the ADA 2026 Standards, cost and access are recognized barriers to optimal therapy. Metformin is generic and inexpensive, typically $4–$10/month at any pharmacy. For Ozempic, Novo Nordisk’s NovoCare program offers a self-pay option at $349/month for 0.25–1 mg doses (verified April 2026) with an introductory offer of $199/month for the first two fills through June 30, 2026. List price is $1,028/month. Telehealth services like WeightLossInjections.com can assist with prior authorization support as part of [$X/month] bundled care ([service detail]).
Should I stop metformin when I start Ozempic?
No, in the vast majority of cases. The ADA 2026 treatment algorithm and the FDA Ozempic clinical study evidence consistently show semaglutide added on top of metformin, not replacing it. Metformin handles basal glucose suppression and insulin sensitization in ways that complement rather than duplicate Ozempic’s mechanism. The only scenario where a provider might reconsider metformin continuation is in advancing CKD (eGFR approaching 30) or a significant GI intolerance that cannot be managed with dose or formulation adjustments.
This article is for educational purposes only and is not a substitute for professional medical advice, diagnosis, or treatment. WeightLossInjections.com’s medical team reviews content quarterly; last medical review: April 2026. Always consult your licensed healthcare provider before making any changes to your medication regimen.

