Semaglutide Plasma Level Timeline After Missed Dose
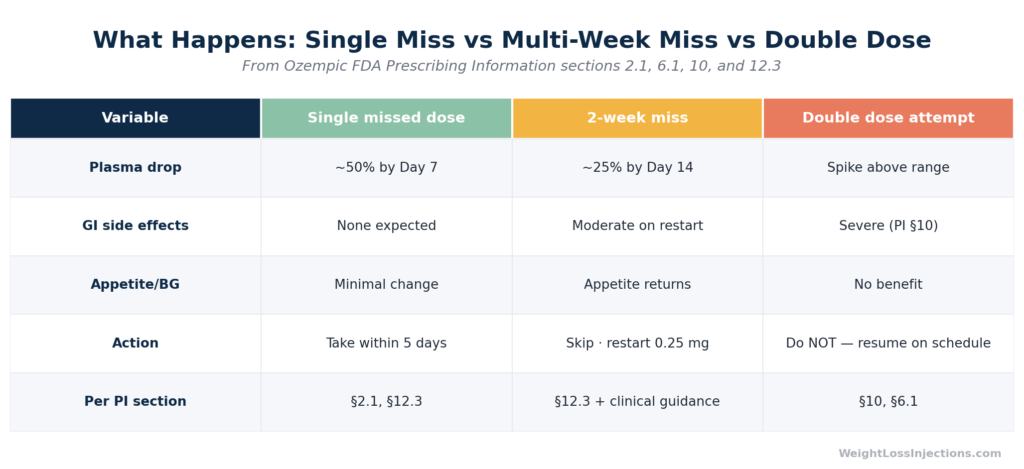
Missing a single Ozempic dose rarely causes dramatic symptoms. Semaglutide’s half-life of approximately one week means levels drop slowly, but if you miss your scheduled injection, the official FDA prescribing information rule is clear: take it within 5 days, or skip it entirely and resume your next scheduled dose. Never double up. If you’ve missed two or more weeks, consult your provider before resuming, you may need to restart at a lower titration dose.
What Happens If You Miss an Ozempic Dose?
Ozempic (semaglutide) is not a drug you take and feel within the hour. It works gradually, building plasma concentration over weeks of consistent once-weekly dosing. That mechanism is also what protects you when you miss a dose.
The Half-Life Explanation
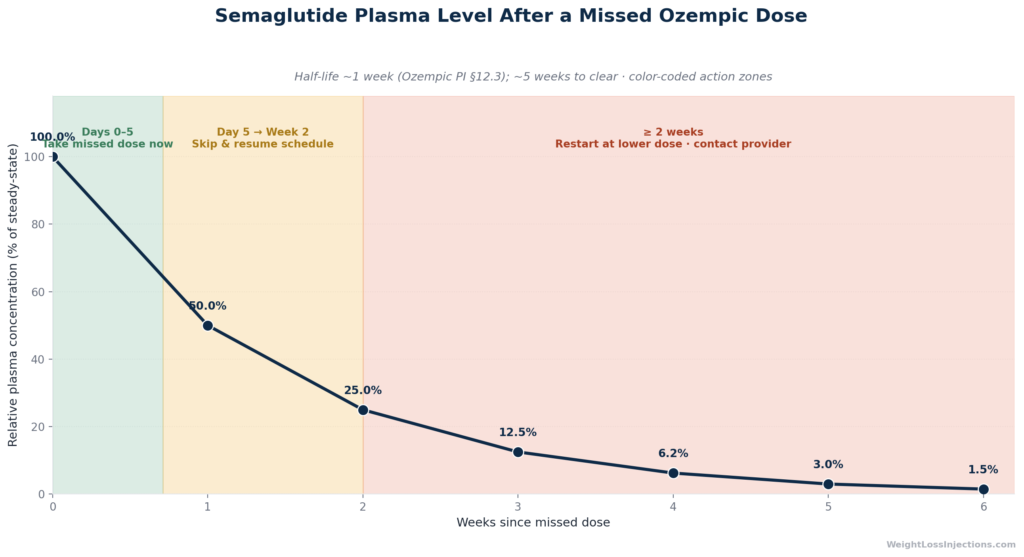
Per Ozempic prescribing information section 12.3 (Novo Nordisk, 2025), semaglutide has an elimination half-life of approximately one week. This means that after one week without a dose, approximately 50% of the drug’s concentration remains in your system. Two weeks out, roughly 25% remains. The same PI states explicitly: “semaglutide will be present in the circulation for about 5 weeks after the last dose.”
In practical terms, missing one weekly injection produces only a modest dip in plasma concentration, not a cliff drop. The drug does not clear your system within days the way a short-acting medication would. Steady-state exposure, per the same Ozempic PI §12.3, is achieved after 4 to 5 weeks of once-weekly administration, meaning one missed injection near steady state creates a partial, not total, interruption of coverage.
Blood Sugar and Appetite Effects
For patients using Ozempic for type 2 diabetes, a missed dose may cause a modest rise in blood glucose, particularly in the days following the miss. The degree depends on how many other medications you’re taking, your baseline glycemic control, and how long you’ve been at steady-state levels. Mayo Clinic’s semaglutide guidance recommends diabetic patients monitor blood sugar more closely during any dose interruption.
For patients using Ozempic off-label for weight management, the most common effect of a missed dose is the gradual return of appetite and what patients often describe as “food noise”, the intrusive thoughts about eating that semaglutide suppresses. This return does not happen overnight, given the residual concentration in circulation, but most patients notice increased hunger by days 5–7 post-miss if they have not yet taken a replacement dose. GoodRx pharmacist guidance notes that a single missed dose is unlikely to have significant effects, while multiple misses reduce effectiveness further and may allow early weight regain.
Official Instructions: Within 5 Days or Skip?
The Ozempic prescribing information section 2.1 (Novo Nordisk, 2025) is unambiguous:
“If a dose is missed, administer OZEMPIC as soon as possible within 5 days after the missed dose. If more than 5 days have passed, skip the missed dose and administer the next dose on the regularly scheduled day. In each case, patients can then resume their regular once-weekly dosing schedule.”
This same rule is confirmed by Mayo Clinic and GoodRx.
Days-Late Decision Matrix
Use this table based on a Monday scheduled injection day:
| Days Since Missed Dose | Day of Week (Monday Schedule) | Action | Resume Schedule |
|---|---|---|---|
| Day 1 | Tuesday | Take missed dose now | Continue next Monday as normal |
| Day 2 | Wednesday | Take missed dose now | Continue next Monday as normal |
| Day 3 | Thursday | Take missed dose now | Continue next Monday as normal |
| Day 4 | Friday | Take missed dose now | Continue next Monday as normal |
| Day 5 | Saturday | Take missed dose now (last window) | Continue next Monday as normal |
| Day 6+ | Sunday onward | Skip it entirely | Take next dose Monday as normal — do not double up |
One important nuance: the Ozempic PI §2.1 also permits changing your injection day, as long as at least 48 hours (2 days) have passed since your last dose. So if you realize mid-week that you want to shift your dosing day permanently, you can do so without requiring a full week’s gap.
Side Effects of Missing Ozempic Doses
Single Missed Dose
A single missed injection, handled correctly within the 5-day window, produces no distinct side effects in most patients. Because semaglutide’s residual half-life keeps meaningful drug concentrations circulating for days after the miss, the physiological interruption is gradual. GoodRx notes there are “no traditional withdrawal symptoms” with GLP-1 receptor agonists.
For T2D patients, self-monitoring blood glucose a few times per day during the miss is prudent but not urgent unless your diabetes is poorly controlled or you are on insulin (Mayo Clinic, 2026).
Multiple Misses (1–2+ Weeks)
Missing two or more consecutive doses is where meaningful clinical effects begin to accumulate. Blood sugar may rise substantially in T2D patients. For weight management patients, the return of appetite becomes progressively more noticeable as plasma levels continue falling by half every seven days.
The larger risk at this stage is restarting at your current dose without a protocol adjustment. Per GoodRx pharmacist guidance, restarting after multiple missed doses may increase side effects such as nausea and diarrhea, particularly at higher doses, because GI tolerance built during initial titration begins to wane after extended absence. The longer the interruption, the more the stomach “forgets” its adapted state.
Risks of Double Dosing
Double dosing is not a recovery strategy, it is an overdose scenario. Taking two doses at once (or two doses within a very short window) dramatically spikes semaglutide concentrations above the therapeutic range, overwhelming the GI system’s capacity to adapt.
The Ozempic PI §10 Overdosage section (Novo Nordisk, 2025) notes that in an overdose event, supportive treatment should be initiated based on clinical signs and symptoms, and that “a prolonged period of observation and treatment for these symptoms may be necessary, taking into account the long half-life of OZEMPIC of approximately 1 week.” In trial populations, GI adverse reactions, nausea, vomiting, diarrhea, were substantially more common at higher doses: vomiting occurred in 9.2% of patients at 1 mg and was noted more frequently at 2 mg (FDA Ozempic PI §6.1, 2025). A self-administered double dose can produce similar concentration spikes.
The bottom line is the same regardless of intent: do not take two Ozempic doses to compensate for a missed one. The FDA label, Mayo Clinic, and GoodRx all explicitly prohibit this.
Missed Dose Comparison Table
Restarting After Multiple Missed Doses
If you’ve missed two to four weeks of Ozempic injections, resuming at your current dose, especially if that’s 1 mg or 2 mg, carries meaningful risk of severe GI side effects. The physiological tolerance your body built during the original titration process is not permanent; it begins degrading within weeks of the drug clearing your system.
The Lower-Dose Restart Protocol
Clinical guidance from GoodRx recommends that patients who miss two or more consecutive doses contact their prescriber, who may temporarily lower the dosage for several weeks before resuming their previous maintenance dose. This mirrors the original titration logic outlined in the FDA Ozempic PI §2.2: starting at 0.25 mg for four weeks to allow GI adaptation before escalating.
In practical terms, most providers will recommend restarting at 0.25 mg or 0.5 mg, effectively re-running the initial titration ramp, before returning to your previous therapeutic dose. The Novo Nordisk Ozempic dosing page and secondary clinical sources including Healthline’s semaglutide guide support this restart-at-lower-dose approach after multi-week interruptions.
Contact Your Provider Protocol
Reach out to your prescriber or telehealth provider before resuming if:
- You have missed two or more consecutive doses
- You are unsure whether your current pen still has viable medication (check the expiration date on the pen — stored properly, an open pen is usable for up to 56 days at room temperature up to 86°F, per the Ozempic PI storage section)
- You experienced significant nausea or vomiting on your previous dose and are concerned about restarting
- You are managing T2D and your blood sugar has been elevated during the interruption
Your prescriber can adjust the restart dose, confirm pen viability, and update your titration plan accordingly. Do not guess.
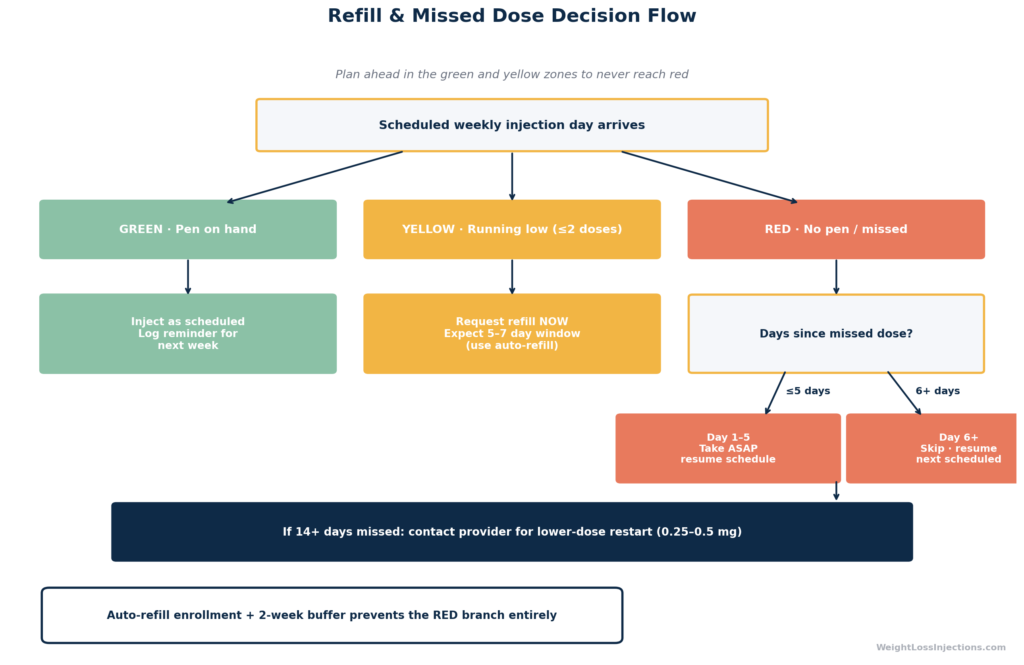
Prevention Tips for Telehealth Patients
The most common reason telehealth patients miss doses is not forgetfulness, it is supply disruption. Understanding the current supply landscape and building redundancy into your refill process is the most reliable prevention strategy.
Current Supply Status
The FDA declared the Ozempic shortage resolved on February 21, 2025. Since that date, compounded versions from 503A and 503B pharmacies have faced compliance deadlines (503A: April 22, 2025; 503B: May 22, 2025), reducing the compounded supply pipeline. Brand-name Ozempic is commercially available, with a list price of $1,028/mo and NovoCare self-pay programs offering $349/mo for 0.25–1 mg doses and $499/mo for 2 mg (Novo Nordisk NovoCare program). Demand for brand-name product remains high, however, and retail pharmacy stock can still vary by geography.
Reminder and Auto-Refill Strategies
- Set a weekly phone alarm for your injection day. Many patients link it to a consistent weekly habit — Sunday dinner, Monday morning — to anchor the injection in routine.
- Use pharmacy auto-refill. Request a 90-day supply where possible. Most pharmacies and telehealth platforms allow auto-refill enrollment that ships your next pen before the current one runs out.
- Request refills early. Ozempic pens hold four doses (at 0.5 mg, 1 mg, or 2 mg). Request your refill at the two-dose mark — not when you’re down to the last dose — to account for processing and shipping delays.
- Know your telehealth provider’s escalation path. If your prescription is managed via telehealth, confirm how quickly your provider can issue an emergency refill authorization if your pharmacy is out of stock.
Our take at WeightLossInjections.com: Supply-chain gaps are the hidden culprit behind most multi-week Ozempic misses. At WeightLossInjections.com, our telehealth service model includes proactive refill coordination so you’re not scrambling when a pen runs out. A licensed provider reviews your case within 24 hours and manages your prescription for [$X/month] including [service detail]. Start your intake at WeightLossInjections.com — consistent dosing is where consistent results come from.
Ozempic Refill Timeline Flowchart
When to Call Your Doctor
Not every missed dose requires a phone call, but some situations do:
- Two or more consecutive missed doses: Your restart protocol needs to be tailored to how long you’ve been off the medication.
- Severe nausea, vomiting, or diarrhea when restarting after a miss — particularly if you resumed at your previous maintenance dose. Persistent vomiting or inability to keep liquids down warrants same-day contact (FDA Ozempic PI §6.1).
- Significant blood sugar elevation in T2D: If your glucose has been running high for several days following a miss, your provider may want to assess whether a temporary insulin bridge is warranted.
- Any uncertainty about timing or dose: If you genuinely cannot remember whether you took your last dose, your provider can help you reconstruct the timeline based on your refill and administration records.
- Changing medications: If you are starting another medication that affects GI motility or blood sugar, your prescriber needs to know about any Ozempic dose interruptions before adjusting your regimen.
The Mayo Clinic semaglutide page advises contacting your provider any time you are uncertain about how to restart treatment after a gap. That is the right default.
Frequently Asked Questions
What if I miss Ozempic by 3 days?
Take your missed dose as soon as you realize, Day 3 falls well within the official 5-day window from the Ozempic prescribing information §2.1 (Novo Nordisk, 2025). After taking the missed dose, resume your normal weekly schedule on the same day of the week you always inject. Your next dose should be on its usual day, not earlier. No dose reduction or special restart protocol is needed for a single 3-day delay.
Does missing one Ozempic dose cause weight gain?
Unlikely in the immediate term. Because semaglutide has a half-life of approximately one week, roughly half the drug’s concentration remains at Day 7 after the miss, Ozempic PI §12.3 confirms the drug is “present in the circulation for about 5 weeks after the last dose.” One missed injection produces a partial, gradual dip in appetite suppression rather than an abrupt loss of effect. Most patients notice increased hunger starting around Days 5–7 post-miss, per GoodRx clinical guidance, but meaningful weight regain requires multiple consecutive missed doses, not one.
Can I take a double dose of Ozempic to catch up?
No. Taking two doses at once is explicitly prohibited by the Ozempic prescribing information §2.1 and confirmed by Mayo Clinic and GoodRx. Doubling the dose creates a concentration spike well above the therapeutic range, which the Ozempic PI §10 overdosage section notes requires supportive clinical management given semaglutide’s prolonged half-life. The most common consequence is severe nausea and vomiting, the FDA PI §6.1 shows vomiting rates of 9.2% at 1 mg in controlled trials; a self-administered double dose can exceed that spike. There is no catch-up benefit, skip the missed dose if you are outside the 5-day window and resume normally.
How long does Ozempic stay in your system after a missed dose?
Approximately 5 weeks from the last dose administered. Per the Ozempic PI §12.3 pharmacokinetics section, semaglutide’s elimination half-life is approximately one week, meaning it takes roughly five half-lives — five weeks — to fall below detectable levels. The concentration curve is: ~50% at Day 7, ~25% at Day 14, ~12.5% at Day 21, ~6% at Day 28, ~3% at Day 35 (clinically negligible). This extended presence is why a single missed dose has only modest clinical impact, and also why restarting requires care, residual drug is still present even if it’s no longer at therapeutic levels.
How do I restart Ozempic after missing 3 weeks?
After a 3-week gap, consult your provider before taking any additional doses. At three weeks post-miss, approximately 12.5% of your previous steady-state concentration remains, per the Ozempic PI half-life data. Physiological GI tolerance established during your original titration may have partially reset. Most providers will recommend restarting at a lower dose, typically 0.25 mg or 0.5 mg, for two to four weeks before returning to your previous maintenance dose, mirroring the original titration protocol (GoodRx GLP-1 restart guidance). Do not self-restart at 1 mg or 2 mg after a multi-week interruption without clinical guidance. At WeightLossInjections.com, our providers are available for same-week consults for patients navigating restart scenarios.
This article is for educational purposes only and does not constitute medical advice. WeightLossInjections.com’s medical team reviews content quarterly; last medical review: April 2026. If you are considering Ozempic or any GLP-1 medication, consult a licensed provider about your individual history and circumstances.

