Three Ozempic Pens Side-by-Side Infographic
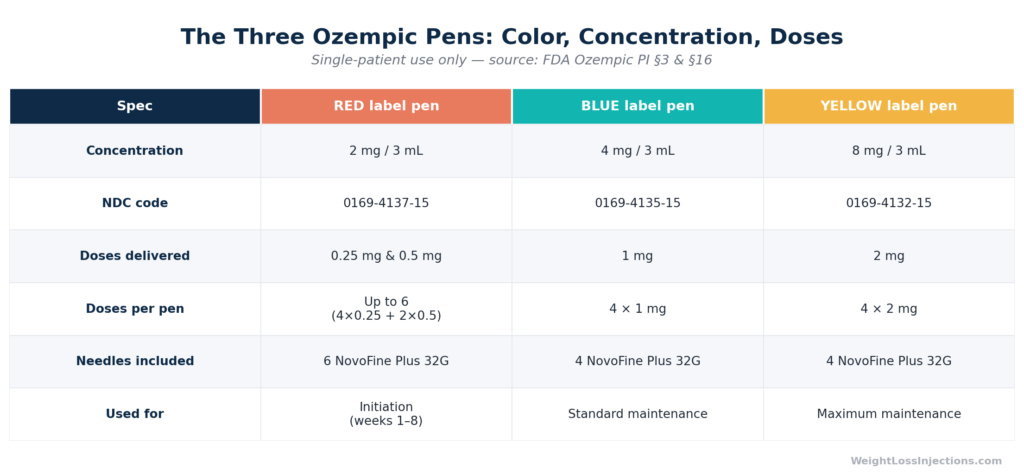
Ozempic comes in three pen configurations: red-label (2 mg/3 mL, 0.25 mg and 0.5 mg doses), blue-label (4 mg/3 mL, 1 mg doses), and yellow-label (8 mg/3 mL, 2 mg doses). All pens are single-patient use only.
The FDA-labeled titration starts at 0.25 mg weekly for four weeks, escalates to 0.5 mg, then optionally to 1 mg and 2 mg, minimum four weeks at each step. Compounded semaglutide is no longer legally permissible at most compounding pharmacies and outsourcing facilities following the FDA’s shortage-resolution order of February 21, 2025.
What Are Ozempic Pens?
Ozempic (semaglutide injection) is dispensed exclusively as a prefilled, multi-dose subcutaneous injection pen made by Novo Nordisk. There is no vial formulation at retail; the pen is the only approved delivery format for branded Ozempic. Per FDA Ozempic Prescribing Information §16, every pen is single-patient use only, sharing a pen between patients is prohibited, even with a new needle, because backflow contamination of the pen mechanism can transmit bloodborne pathogens.
Each pen includes NovoFine Plus 32-gauge, 4 mm needles; a fresh needle must be used for every injection.
The Three Pen Configurations
Novo Nordisk produces three distinct Ozempic pen strengths, differentiated by label color and drug concentration. The same dial position produces a different milligram dose depending on which pen is loaded, always confirm your pen color matches your prescription.
| Pen Color | Concentration | Doses Delivered | Doses Per Pen | Needles | NDC |
|---|---|---|---|---|---|
| Red | 2 mg / 3 mL | 0.25 mg or 0.5 mg | Up to 6 (4×0.25 + 2×0.5 mg) or 4×0.5 mg | 6 NovoFine Plus | 0169-4137-15 |
| Blue | 4 mg / 3 mL | 1 mg | 4 doses | 4 NovoFine Plus | 0169-4135-15 |
| Yellow | 8 mg / 3 mL | 2 mg | 4 doses | 4 NovoFine Plus | 0169-4132-15 |
Source: FDA Ozempic PI §3 and §16; Novo Nordisk Ozempic Instructions for Use; NovoMedLink Ozempic pen page.
The red pen’s flexibility is often misunderstood. It can deliver both 0.25 mg and 0.5 mg from the same cartridge, the dose selector switches between both settings. A patient following the standard titration (weeks 1–4 at 0.25 mg, then weeks 5–8 at 0.5 mg) can use the same red pen for the first six weeks: four injections at 0.25 mg plus two injections at 0.5 mg exhaust the pen’s 2 mg supply. Patients who skip the 0.25 mg initiation phase and begin at 0.5 mg get four doses per red pen, a one-month supply.
Ozempic Dosing Schedule: Weeks 1 Through 20
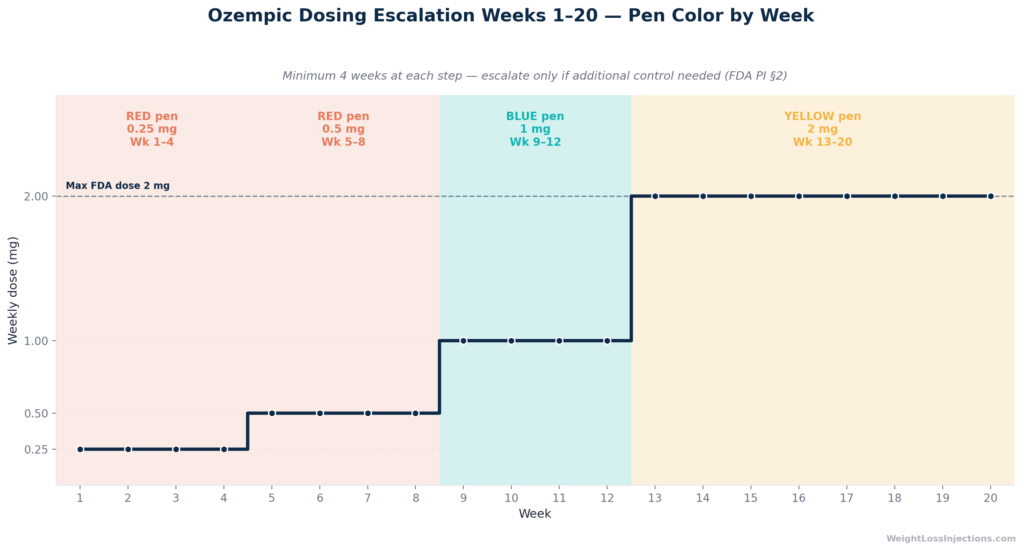
The FDA-labeled dosing ladder exists to reduce gastrointestinal side effects by progressively adapting the GI tract to semaglutide’s gastric-emptying effect before reaching a therapeutic dose. Per FDA Ozempic PI §2, the minimum duration at each level before escalation is four weeks. Escalation is conditional on clinical need, not automatic.
Ozempic Dosing Escalation Timeline Weeks 1–20 with Pen Color by Week
Weeks 1–4: 0.25 mg (Red Pen)
Per FDA Ozempic PI §2, the 0.25 mg starting dose is not intended for glycemic control, it is a tolerance-building step only. It minimizes the nausea and vomiting that occur when semaglutide slows gastric emptying. Do not escalate early. Use the red-label pen at this phase.
Weeks 5–8: 0.5 mg (Red Pen)
The first therapeutic dose. In the SUSTAIN-1 trial (Diabetes Care, 2017), semaglutide 0.5 mg produced a mean 4.5% body weight reduction at 30 weeks in patients with type 2 diabetes. GI side effects typically peak at the week-5 step-up and then decline over two to three weeks. Some patients maintain 0.5 mg as their long-term dose if adequate glycemic or clinical benefit is achieved, per FDA Ozempic PI §2, escalation beyond 0.5 mg requires clinical rationale, not just the passage of time.
Weeks 9–12 (Optional): 1.0 mg (Blue Pen)
Switch to the blue-label pen when escalating to 1 mg. In SUSTAIN-1, semaglutide 1.0 mg produced a mean 6.0% body weight reduction at 30 weeks (SUSTAIN-1, Diabetes Care, 2017). The blue pen provides exactly four 1 mg doses, one month of supply.
CKD note: For patients with type 2 diabetes and chronic kidney disease (the FLOW trial indication, approved January 28, 2025), the FDA Ozempic PI §2 specifies a maintenance ceiling of 1.0 mg weekly, escalation to 2 mg is not indicated in this population.
Weeks 13+ (Optional): 2.0 mg (Yellow Pen)
The maximum FDA-labeled dose. Switch to the yellow-label pen. The SUSTAIN FORTE trial showed an additional ~0.23% HbA1c reduction and modest incremental weight loss versus 1.0 mg (SUSTAIN FORTE, The Lancet Diabetes & Endocrinology, 2021). The yellow pen provides four 2 mg doses, one month. GI adverse events increase modestly at this dose, per FDA Ozempic PI §6.
Dosing Ladder Summary
| Weeks | Dose | Purpose | Pen | Pens/Month |
|---|---|---|---|---|
| 1–4 | 0.25 mg weekly | GI tolerance; not therapeutic | Red | 1 (partial) |
| 5–8 | 0.5 mg weekly | First therapeutic dose | Red | 1 |
| 9–12 | 1.0 mg weekly | Optional escalation | Blue | 1 |
| 13+ | 2.0 mg weekly | Optional max dose | Yellow | 1 |
Source: FDA Ozempic Prescribing Information §2; NovoMedLink Ozempic Dosing and Administration.
How to Use an Ozempic Pen: Step-by-Step
The following steps reflect the Novo Nordisk Ozempic Instructions for Use and FDA Ozempic PI §2. Read the full IFU included with your pen before your first injection.
1. Prepare. Wash hands. Remove the pen from the refrigerator 15–30 minutes before injecting to reduce injection-site stinging. Inspect the solution through the pen window, it should be clear and colorless to slightly yellow, with no particles. Per FDA Ozempic PI §16, do not warm the pen in hot water or a microwave.
2. Attach a new needle. Wipe the rubber stopper with an alcohol swab. Screw a new NovoFine Plus needle clockwise until secure. Remove the outer needle cap (save it for recapping) and the inner needle cap. Never reuse a needle, per Novo Nordisk IFU, reused needles risk blocking the flow path and causing inaccurate dosing.
3. Perform a flow check (first use of each new pen only). Turn the dose selector to the flow check symbol. Hold the pen needle-up and press the dose button until the counter returns to 0. A small drop should appear at the needle tip. If no drop appears after two attempts, replace the needle and try again; if still no flow, contact your pharmacy per Novo Nordisk IFU guidance.
4. Select your dose. Dial to your prescribed dose and confirm the number in the dose window matches your prescription exactly.
5. Inject. Approved sites per FDA Ozempic PI §2: abdomen (≥2 inches from the navel), upper thigh, or upper arm (outer surface). Rotate sites weekly, injecting the same location repeatedly causes lipohypertrophy, which impairs drug absorption. Clean the site with an alcohol swab, insert the needle at 90°, press and hold the dose button until the counter reads 0, then count 6 seconds before withdrawing. The 6-second hold ensures the full dose is delivered, per Novo Nordisk IFU.
6. Recap and dispose. Recap the needle using a one-handed technique, unscrew it from the pen, and discard it in an FDA-cleared sharps container. Replace the pen cap. Log your injection date and site.
Storage and Handling
Correct storage directly affects semaglutide potency, degradation can occur with temperature excursions without any visible change in the solution’s appearance.
Per FDA Ozempic PI §16 and Novo Nordisk IFU:
| Status | Temperature | Limit |
|---|---|---|
| Unused (refrigerated) | 36–46°F (2–8°C) | Until expiration date |
| In-use (after first injection) | Up to 77°F (25°C) — room temp | ≤56 days from first use |
| All pens — freeze risk | Below 32°F (0°C) | Never freeze — discard if frozen |
Do not store the pen with a needle attached after injecting — a capped needle left in place can vent air into the cartridge and cause dosing errors.
Travel tips for telehealth patients: Ozempic ships in insulated packaging with refrigerant gel packs. Refrigerate immediately on receipt — do not leave in a mailbox in temperatures above 77°F. For air travel, carry the pen in your carry-on (TSA permits injectable medications without a size limit). An in-use pen can travel at room temperature for the duration of its 56-day window, as long as it stays below 77°F and out of direct sunlight. Keep it out of hot cars and gloveboxes.
Missed Doses
Per FDA Ozempic PI §2:
- Within 5 days of the missed injection: take the dose as soon as you remember.
- More than 5 days have passed: skip the missed dose entirely. Do not double-dose. Resume the regular weekly schedule.
Set a consistent injection day, same time each week, to minimize missed doses. A weekly phone reminder is a straightforward safeguard.
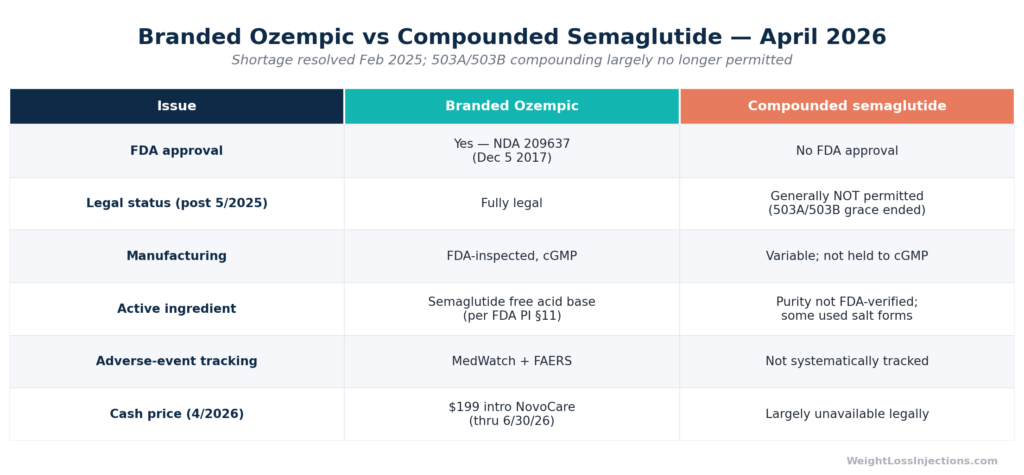
Ozempic Pens vs. Compounded Semaglutide
The Shortage Period and Its End
From 2022 through early 2025, FDA maintained semaglutide on its official drug shortage list. During that window, federal law (21 U.S.C. § 503A and § 503B) permitted compounding pharmacies and outsourcing facilities to produce semaglutide products as shortage-status alternatives, under FDA compounding guidance.
On February 21, 2025, FDA declared the semaglutide shortage resolved (FDA semaglutide shortage resolution order, Feb. 21, 2025). Once a shortage is resolved, the legal basis for compounding commercially available drug products lapses. FDA established grace periods before enforcement:
- 503A compounding pharmacies (patient-specific): Grace period ended April 22, 2025, per FDA 503A enforcement guidance.
- 503B outsourcing facilities (bulk supply): Grace period ended May 22, 2025, per FDA outsourcing facility guidance.
After those dates, compounding semaglutide products for general dispensing is generally not permitted. FDA has issued warning letters and pursued injunctions against compounders who continued production beyond the enforcement deadlines, per FDA 2025 compounding enforcement actions.
Salt Form Concerns
A specific safety issue during the shortage period: many compounders used semaglutide acetate or semaglutide sodium salt forms rather than the semaglutide free acid base in FDA-approved Ozempic. Bioequivalence to branded Ozempic has not been established for these salt forms, per FDA safety communication on compounded semaglutide salt forms (2024). Multiple adverse event reports linked to compounded products, including dosing errors, injection-site infections, and GI hospitalizations, were reviewed by FDA, per FDA MedWatch compounded semaglutide safety reports (2023–2025).
Branded Ozempic vs. Compounded Semaglutide Comparison Table
Our take at WeightLossInjections.com: The compounded semaglutide window is legally closed for the vast majority of patients. If a telehealth provider is still marketing “compounded semaglutide” at a discount in mid-2025 or later, it is operating outside FDA-sanctioned boundaries in virtually all circumstances, scrutiny is warranted. The legitimate self-pay path is Novo Nordisk’s NovoCare program ($199/mo introductory through June 30, 2026; $349–$499/mo ongoing) or equivalent GoodRx pricing. WeightLossInjections.com offers supervised access to FDA-approved Ozempic starting at [$X/month], including [service detail]. No compounded products. No regulatory shortcuts.
Pen Pricing and Cash-Pay Options (April 2026)
Ozempic’s list price is $1,028/mo as of April 2026, per GoodRx Ozempic, but most patients pay substantially less through insurance or direct-pay programs:
| Pathway | Dose | Monthly Cost |
|---|---|---|
| Commercial insurance + Novo Nordisk savings card | Any dose | As low as $25 |
| NovoCare — intro offer (new patients, through 6/30/26) | 0.25 or 0.5 mg | $199 |
| NovoCare — ongoing self-pay | 0.25, 0.5, or 1 mg | $349 |
| NovoCare — ongoing self-pay | 2 mg | $499 |
| List price (no program, no insurance) | Any | $1,028 |
Sources: Ozempic.com savings page; GoodRx Ozempic; verified April 2026. Note: Novo Nordisk announced a $675/mo list price reset for all Ozempic dosage forms beginning in 2027 (Novo Nordisk 2026 pricing announcement); 2026 pricing is unchanged. Government beneficiaries (Medicare, Medicaid, TRICARE, VA) are excluded from the savings card and NovoCare self-pay programs.
Side Effects at a Glance
Ozempic’s GI side-effect profile is the most clinically relevant factor in dose management and timing of escalation. The following rates are from FDA Ozempic PI §6 (pooled placebo-controlled trials across the SUSTAIN program):
| Adverse Effect | 0.5 mg | 1 mg | Placebo |
|---|---|---|---|
| Nausea | 15.8% | 20.3% | 6.1% |
| Vomiting | 5.0% | 9.2% | 2.3% |
| Diarrhea | 8.5% | 8.8% | 1.9% |
| Constipation | 5.0% | 3.1% | 1.5% |
Rates decline over time as GI tolerance develops, typically after 4–8 weeks at each new dose level. The four-week minimum between escalation steps is specifically designed to allow this physiological adaptation window, rushing the escalation schedule is the most common driver of GI-related discontinuation. Eating smaller, lower-fat meals and staying well-hydrated during the first few weeks at a new dose significantly reduces symptom burden in practice, though these strategies are not from controlled trial data. Cardiovascular safety was established in the SUSTAIN 6 trial (NEJM, Marso et al., 2016), a 3,297-patient trial that demonstrated a 26% relative risk reduction in cardiovascular death, nonfatal MI, or nonfatal stroke versus placebo in high-CV-risk T2D patients at 0.5–1.0 mg weekly dosing.
Frequently Asked Questions
How many doses are in a red Ozempic pen?
The red-label Ozempic pen (2 mg/3 mL, NDC 0169-4137-15) delivers up to 6 doses across both dose settings: four at 0.25 mg followed by two at 0.5 mg (spanning 6 weeks). Used exclusively at 0.5 mg, it provides 4 doses, one month of supply. Per FDA Ozempic PI §3 and Novo Nordisk IFU, the pen includes 6 NovoFine Plus needles to accommodate both use patterns.
Can I use an Ozempic pen for 56 days after opening?
Yes, with caveats. Per FDA Ozempic PI §16, an in-use pen may be kept at room temperature (up to 77°F / 25°C) for up to 56 days from the date of first use. After 56 days, discard the pen even if solution remains. The pen must never be frozen, exposure below 32°F is grounds for immediate disposal, regardless of remaining solution. The 56-day clock begins with the first injection, whether the pen is stored at room temperature or returned to the refrigerator between doses.
What should I do if I miss an Ozempic dose?
Per FDA Ozempic PI §2: if 5 days or fewer have elapsed since your scheduled injection day, take the missed dose as soon as possible. If more than 5 days have passed, skip the missed dose entirely and resume your regular weekly schedule. Never take two doses to make up for a missed one. A single missed injection will not materially disrupt steady-state semaglutide levels, the drug’s ~7-day half-life means plasma levels decline slowly over a missed week.
What is the difference between the red, blue, and yellow Ozempic pens?
The three pens differ in drug concentration, available dose settings, and doses per pen. The red pen (2 mg/3 mL) is the titration-phase pen, delivering 0.25 mg and 0.5 mg doses for the first 8 weeks. The blue pen (4 mg/3 mL) delivers 1 mg per injection, 4 doses per pen, for the optional first escalation step. The yellow pen (8 mg/3 mL) delivers 2 mg per injection, 4 doses per pen, for the maximum FDA-labeled dose. All three are single-patient use only per FDA Ozempic PI §2; sharing a pen between individuals is prohibited regardless of needle changes.
How should I store Ozempic pens during travel?
Per FDA Ozempic PI §16: unused pens must be refrigerated at 36–46°F, use an insulated medication case with gel packs if traveling without refrigeration access for more than a few hours. An in-use pen can be kept at room temperature (up to 77°F) for the duration of its 56-day window. Carry pens in your carry-on luggage, do not check them in baggage, where cargo hold temperatures are uncontrolled. TSA permits injectable medications in carry-on bags without the standard 3.4 oz liquid rule. For international travel, a prescription label or physician note is advisable per Novo Nordisk IFU guidance. Keep pens out of direct sunlight and away from hot car surfaces.
This article is for informational purposes only and does not constitute medical advice. Dosing decisions, including initiation, escalation, and hold, are your prescribing clinician’s determination. Storage instructions and compounding legal status reflect FDA guidance as of April 2026 and are subject to change. WeightLossInjections.com editorial team verifies content quarterly; last verified April 2026.

