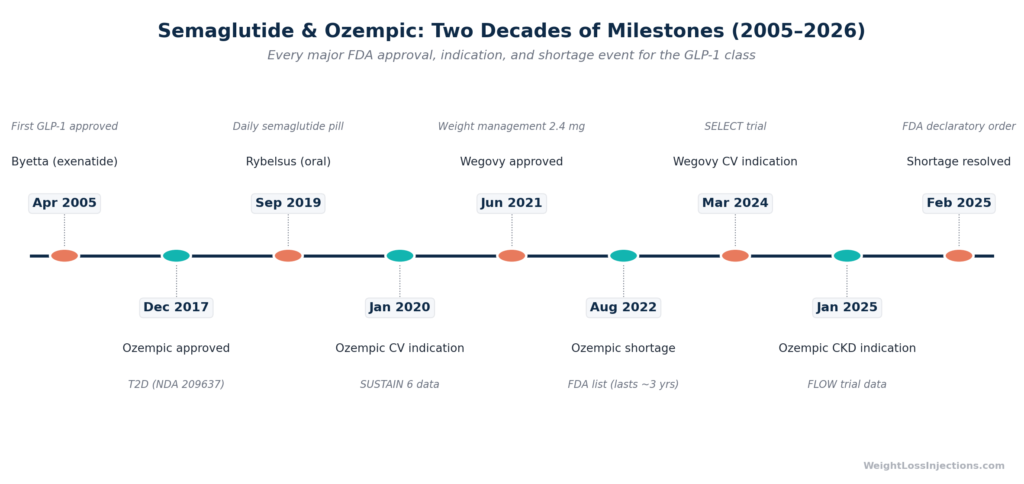
Ozempic (semaglutide injection) has been on the U.S. market since early 2018, following FDA approval on December 5, 2017 for type 2 diabetes. In the eight years since, the label expanded to include cardiovascular risk reduction (January 2020), a higher 2 mg dose (March 2022), and kidney disease protection (January 2025). Its close relative Wegovy — same molecule, higher dose — was approved for chronic weight management in June 2021. The full history runs from a Gila monster in the 1990s to one of the most-prescribed drugs in the United States.
Ozempic & semaglutide milestone timeline
The GLP-1 backstory: from Gila monster venom to semaglutide
To understand Ozempic’s timeline, you need to understand where the whole drug class came from.
In the early 1990s, Dr. John Eng at the Veterans Affairs Medical Center in the Bronx discovered a hormone in Gila monster venom called exendin-4, a peptide that mimicked the human gut hormone GLP-1 (glucagon-like peptide-1) but degraded far more slowly in the body (VA Research). A synthetic version became exenatide (Byetta), which the FDA approved in April 2005 as the first GLP-1 receptor agonist for type 2 diabetes (PMC / Baylor University Medical Center, 2006). The proof of concept, that a GLP-1 agonist could lower blood sugar and incidentally reduce weight, sent pharmaceutical companies back to their labs.
Novo Nordisk had already been developing liraglutide (Victoza), a once-daily GLP-1 agonist, approved by the FDA in January 2010. The next step was a once-weekly version. Engineers modified the semaglutide molecule with a fatty acid chain that binds tightly to albumin in the blood, extending its half-life to approximately one week. The result was semaglutide, the active ingredient in Ozempic, Wegovy, and Rybelsus.
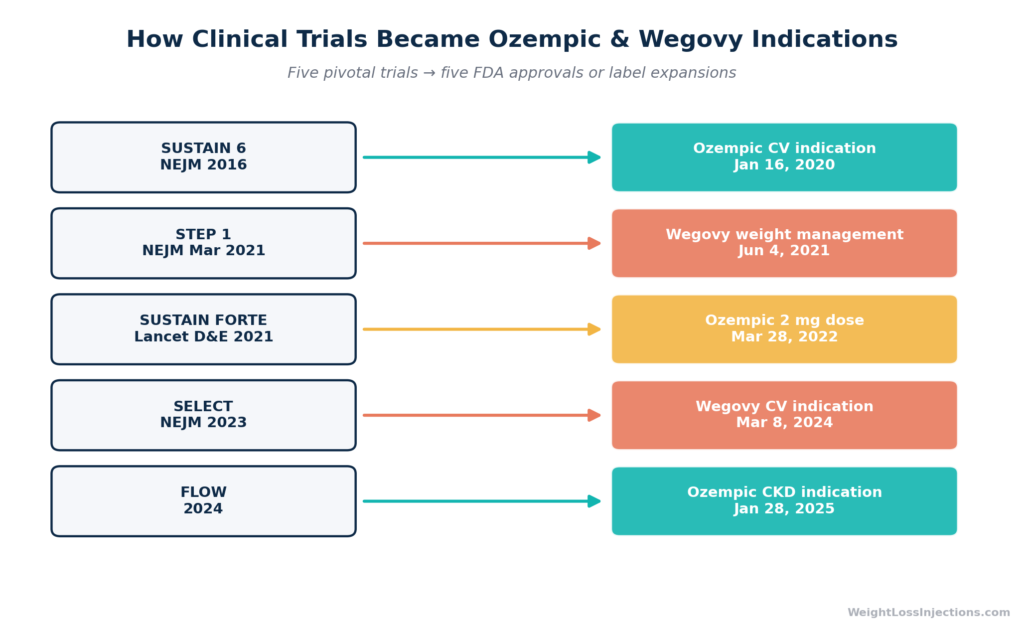
Novo Nordisk’s Phase 3 program for semaglutide injection — the SUSTAIN trials (Semaglutide Unabated Sustainability in Treatment of Type 2 Diabetes) — enrolled more than 8,000 adults with type 2 diabetes across eight trials. The cardiovascular outcomes trial, SUSTAIN 6, published in The New England Journal of Medicine in 2016, enrolled 3,297 adults with type 2 diabetes and established cardiovascular disease. Semaglutide reduced the composite endpoint of CV death, non-fatal heart attack, or non-fatal stroke by 26% vs. placebo (HR 0.74; 95% CI 0.58–0.95) (NEJM, Marso et al., 2016).
December 5, 2017: FDA approves Ozempic for type 2 diabetes
Novo Nordisk submitted NDA 209637 to the FDA in December 2016. After approximately one year of review, the FDA approved Ozempic (semaglutide injection) 0.5 mg and 1 mg on December 5, 2017, as confirmed in the official approval letter (FDA approval letter, NDA 209637, December 5, 2017). Classification: Type 1 – New Molecular Entity, the highest novelty designation.
That same day, Novo Nordisk announced the drug would launch at U.S. pharmacies in Q1 2018 (Novo Nordisk / PR Newswire, December 5, 2017).
Original approved indication: Adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus.
Doses: 0.5 mg and 1 mg weekly, with a 0.25 mg starting dose for titration.
Boxed warning from day one: Potential risk of medullary thyroid carcinoma, based on rodent data. Patients with a personal or family history of MTC or MEN2 should not take Ozempic.
One crucial fact the original approval did not include: any indication for weight loss. That came later, and under a different brand name.
Our take at WeightLossInjections.com: Ozempic’s December 2017 approval is often conflated with Wegovy’s 2021 weight-loss approval in public coverage. They are separate approvals for separate indications. The popular narrative, “Ozempic is a weight-loss drug”, is clinically imprecise. Ozempic is a diabetes drug with consistent weight-loss side effects that was later prescribed widely off-label, until a weight-loss version (Wegovy) could catch up on supply.
2019: Semaglutide goes oral — Rybelsus
On September 20, 2019, the FDA approved Rybelsus (semaglutide tablets, 7 mg and 14 mg), the first oral GLP-1 receptor agonist in U.S. history (Beyond Type 1, September 2019). Rybelsus uses a proprietary absorption enhancer (SNAC) to protect semaglutide from stomach acid. The indication was identical to Ozempic: glycemic control in adults with type 2 diabetes.
Rybelsus has never appeared on the FDA drug shortage list, a fact that would matter enormously when the injectable forms faced severe supply constraints starting in 2022.
January 16, 2020: Cardiovascular risk reduction added to Ozempic’s label
Based on the SUSTAIN 6 data, the FDA approved a supplemental indication for Ozempic on January 16, 2020 (SUPPL-3): to reduce the risk of major adverse cardiovascular events (MACE) in adults with type 2 diabetes and known heart disease (Novo Nordisk / PR Newswire, January 16, 2020; FDA SUPPL-3 label, 2020).
This made Ozempic one of the few diabetes drugs with label-backed cardiovascular benefit, a meaningful clinical distinction as cardiology and endocrinology guidelines began recommending GLP-1 agonists for high-risk patients.
June 2021: Wegovy approval and the weight-loss era
The STEP clinical trial program established semaglutide’s weight-loss evidence base. The landmark STEP 1 trial, published in The New England Journal of Medicine in March 2021, enrolled 1,961 adults with obesity or overweight without type 2 diabetes. After 68 weeks of semaglutide 2.4 mg weekly, mean body weight reduction was –14.9% vs. –2.4% on placebo — a treatment difference of 12.4 percentage points that was unprecedented for a pharmacologic obesity treatment (STEP 1, NEJM, 2021).
On June 4, 2021, the FDA approved Wegovy (semaglutide injection 2.4 mg) under NDA 215256, as an adjunct to a reduced calorie diet and increased physical activity for chronic weight management in adults with BMI ≥30 or ≥27 with at least one weight-related condition (FDA Wegovy approval letter, NDA 215256, June 4, 2021).
Wegovy is chemically identical to Ozempic — the active ingredient is semaglutide in both, but the maximum approved dose (2.4 mg vs. Ozempic’s 2.0 mg) and the weight-management indication are legally distinct. Ozempic is not FDA-approved for weight loss. Wegovy is.
GLP-1 prescription volume growth
2021–2023: The off-label surge and social media wave
Wegovy launched in mid-2021 and almost immediately went into shortage. Ozempic — same drug class, more available — became the de facto weight-loss substitute. Off-label prescribing of Ozempic for people without type 2 diabetes accelerated sharply.
The social media acceleration added an unprecedented demand shock. By late 2022, “#Ozempic” was going viral on TikTok, celebrities were publicly acknowledging semaglutide use, and the drug was being called “the worst-kept secret in Hollywood” (New York Times, November 2022). Real-world prescribing data showed GLP-1 prescriptions for patients with overweight or obesity were 3.4 times higher in the first half of 2023 than the same period in 2021 (ISPOR real-world prescribing analysis, 2023).
The surge created supply strain on a diabetes drug, making it harder for patients with type 2 diabetes to obtain their prescriptions — and triggered regulatory scrutiny of the growing compounded semaglutide market.
Semaglutide shortage timeline
2022–2025: The shortage, resolution, and label expansions
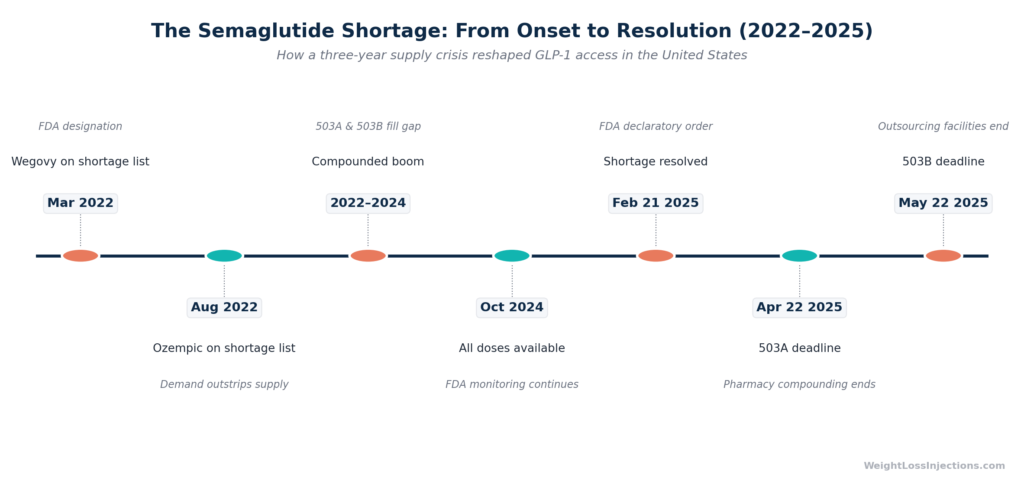
The shortage timeline:
- March 2022: Wegovy added to FDA drug shortage list (FDA Declaratory Order, February 21, 2025)
- August 2022: Ozempic added to FDA drug shortage list (FDA Declaratory Order, February 21, 2025)
- 2022–2024: Compounding pharmacies legally produce semaglutide copies under FDA shortage exemptions; telehealth platforms scale rapidly
- February 21, 2025: FDA issues a formal declaratory order: the semaglutide shortage is resolved. Novo Nordisk is “meeting or exceeding demand for Ozempic and Wegovy” (GoodRx shortage timeline, 2024; Pharmacy Times, February 27, 2025)
The resolution set hard enforcement deadlines: state-licensed pharmacies had until April 22, 2025 to stop compounding semaglutide copies; outsourcing facilities until May 22, 2025.
Concurrent label expansions:
March 28, 2022 — 2 mg dose approved. Based on the SUSTAIN FORTE trial, the FDA approved a 2 mg weekly dose of Ozempic, showing superior A1C reduction vs. 1 mg (–2.1% vs. –1.9%). Ozempic was now available in 0.5 mg, 1 mg, and 2 mg weekly doses (Novo Nordisk / PR Newswire, March 28, 2022; FDA SUPPL-9 label).
September 22, 2023 — Ileus warning added. Post-market reports of intestinal blockage (ileus) prompted a label update (SUPPL-20/21) (FDA SUPPL-20/21, September 22, 2023).
WeightLossInjections.com editorial note: The shortage’s three-year run reshaped the entire GLP-1 landscape. It created the compounded semaglutide industry, accelerated telehealth adoption, and contributed to a wave of litigation when the FDA declared it over. Patients who began treatment on compounded semaglutide faced an abrupt transition period as enforcement deadlines passed in mid-2025.
2024–2026: Cardiovascular proof beyond diabetes, kidney outcomes, and what’s next
March 8, 2024 — Wegovy SELECT cardiovascular indication.
The SELECT trial enrolled 17,604 adults with overweight or obesity and established cardiovascular disease but without type 2 diabetes, testing whether semaglutide could prevent cardiovascular events in people whose only qualifying condition was excess weight. It could. Wegovy reduced the risk of major adverse cardiovascular events (CV death, heart attack, stroke) by 20% vs. placebo (HR 0.80; 95% CI 0.72–0.90; absolute risk reduction 1.5% at 40 months) (FDA press release, March 8, 2024; Novo Nordisk / PR Newswire, March 8, 2024).
This transformed semaglutide from a diabetes drug with beneficial side effects into a proven cardiovascular medicine for the broader overweight population.
January 28, 2025 — Ozempic approved for chronic kidney disease.
Based on the FLOW trial — 3,533 adults with type 2 diabetes and chronic kidney disease, Ozempic received its third indication: to reduce the risk of kidney disease worsening, kidney failure, and cardiovascular death in adults with type 2 diabetes and CKD. FLOW showed a 24% reduction in the composite kidney outcome vs. placebo (Novo Nordisk / Ozempic.com, January 28, 2025; FDA SUPPL-25 label). Ozempic is now the first GLP-1 receptor agonist with a specific renal protection indication.
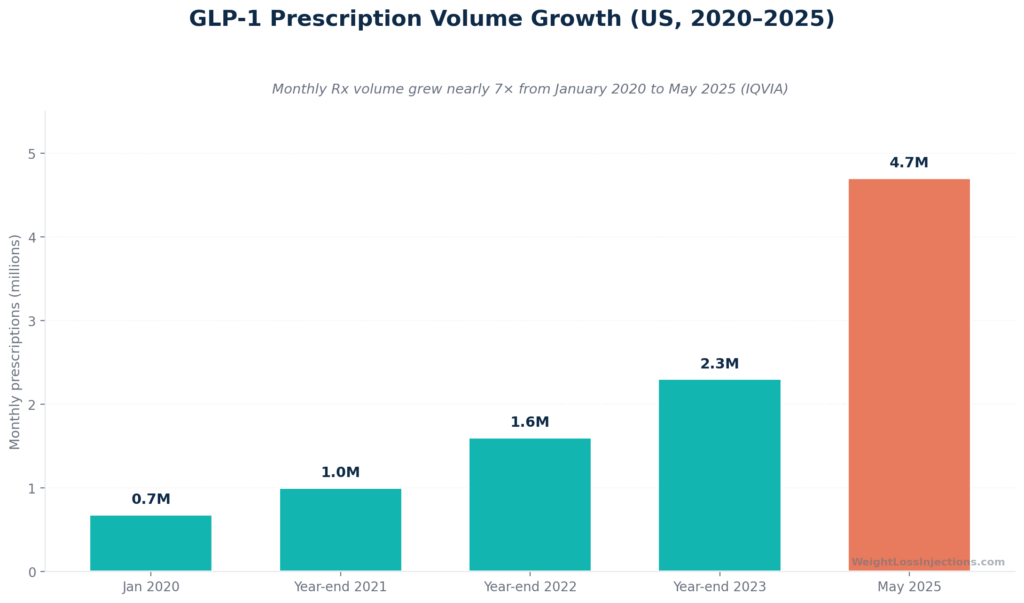
By May 2025, IQVIA data showed GLP-1 commercial prescription volume reaching 4.7 million per month, up from approximately 680,000 in January 2020, a nearly sevenfold increase in five years (IQVIA GLP-1 Impact Report, 2025). GLP-1s now account for 23% of all first-line diabetes therapy, up from 3% in 2021.
Trial-to-indication map
Is Ozempic right for you today?
As of April 2026, Ozempic carries three FDA-approved indications:
- Glycemic control in adults with type 2 diabetes (December 5, 2017)
- Reduction of major cardiovascular events in adults with type 2 diabetes and known heart disease (January 16, 2020)
- Reduction of kidney disease worsening and cardiovascular death in adults with type 2 diabetes and CKD (January 28, 2025)
Weight loss is not on this list. That indication belongs to Wegovy. Off-label prescribing of Ozempic for weight management in adults without type 2 diabetes is legal, but insurance typically will not cover it for that use. If your primary goal is weight loss and you meet Wegovy’s criteria (BMI ≥30, or ≥27 with a weight-related condition), Wegovy is the evidence-backed choice.
At WeightLossInjections.com, our licensed providers evaluate your full medical history to identify the right GLP-1 for your situation. A typical patient pays [$X/month] all-in, which includes the medication, ongoing provider access, and [service detail].
[Check your eligibility — take our 3-minute intake →]
Frequently asked questions
How long exactly has Ozempic been on the market?
Ozempic received FDA approval on December 5, 2017 and launched at U.S. pharmacies in Q1 2018 — making it over eight years old as of 2026 (FDA approval letter, NDA 209637; Novo Nordisk press release, December 5, 2017).
When did Ozempic become available to patients?
Ozempic was FDA-approved December 5, 2017. Novo Nordisk confirmed at the time of approval that U.S. commercial launch would occur in Q1 2018, meaning patients began filling prescriptions in early 2018.
How does Ozempic’s time on the market compare to Wegovy’s?
Ozempic was approved nearly four years before Wegovy. Ozempic: approved December 5, 2017, for type 2 diabetes. Wegovy: approved June 4, 2021, for chronic weight management. Both contain semaglutide, but at different approved maximum doses and with separate FDA approvals for distinct indications (FDA Wegovy approval letter, NDA 215256).
Has Ozempic’s approval expanded beyond its original diabetes indication?
Yes. The original 2017 approval covered only glycemic control. The FDA subsequently approved cardiovascular risk reduction in T2D patients with known heart disease (January 16, 2020) and chronic kidney disease protection (January 28, 2025), based on the SUSTAIN 6 and FLOW trials respectively (Drugs@FDA, NDA 209637).
Is Ozempic considered safe after 8+ years on the market?
The long-term evidence has strengthened. The SELECT trial followed over 17,600 patients for a mean of 40 months, finding a 20% reduction in major cardiovascular events with a consistent safety profile (FDA press release, SELECT approval, March 8, 2024). The known risks — GI side effects, rare pancreatitis, ileus, and the thyroid tumor boxed warning based on rodent data — have been present since 2017 and are reflected in the current prescribing information. Decade-length data continues to accumulate.
Key sources and further reading
- FDA NDA 209637 approval letter, December 5, 2017 — accessdata.fda.gov
- Drugs@FDA: Ozempic complete approval history — accessdata.fda.gov
- Novo Nordisk FDA approval press release, December 5, 2017 — prnewswire.com
- SUSTAIN 6 CV outcomes trial — NEJM, Marso et al., 2016
- Ozempic CV indication press release, January 16, 2020 — prnewswire.com
- FDA Ozempic label with CV indication, January 2020 (SUPPL-3) — accessdata.fda.gov
- STEP 1 trial, semaglutide 2.4 mg in obesity — NEJM, 2021
- FDA Wegovy approval letter, NDA 215256, June 4, 2021 — accessdata.fda.gov
- Ozempic 2 mg approval, March 28, 2022 — prnewswire.com
- FDA label with 2mg dose, March 2022 (SUPPL-9) — accessdata.fda.gov
- FDA Ozempic label update with ileus warning (SUPPL-20/21), September 2023 — accessdata.fda.gov
- FDA declaratory order resolving semaglutide shortage, February 21, 2025 — fda.gov
- Ozempic CKD/FLOW press release, January 28, 2025 — ozempic.com
- FDA label with CKD indication, January 2025 (SUPPL-25) — accessdata.fda.gov
- FDA approves Wegovy for CV risk reduction, March 8, 2024 — fda.gov
- Novo Nordisk Wegovy SELECT press release, March 8, 2024 — prnewswire.com
- IQVIA GLP-1 prescription volume data, 2025 — iqvia.com
- VA Research: Gila monster venom origin story — research.va.gov
- Exenatide (Byetta) FDA approval April 2005 — PMC / Baylor University Medical Center
- Rybelsus FDA approval September 20, 2019 — Beyond Type 1
- ISPOR real-world GLP-1 prescribing data, 2023 — ispor.org
- GoodRx: Semaglutide shortage timeline — goodrx.com
- Pharmacy Times: FDA ends semaglutide shortage, February 27, 2025 — pharmacytimes.com
This article is for educational purposes only and is not a substitute for medical advice. WeightLossInjections.com’s editorial team reviews content quarterly. Last medical review: April 2026. If you are considering Ozempic or any GLP-1 therapy, consult a licensed provider about your individual medical history and treatment goals.

